Chapter 27: Q6P (page 767)
Why is it less desirable to wash AgCl precipitate with aqueous NaNO3 than with HNO3 solution?
Short Answer
The AgCI precipitate would be more desirable than with
Learning Materials
Features
Discover
Chapter 27: Q6P (page 767)
Why is it less desirable to wash AgCl precipitate with aqueous NaNO3 than with HNO3 solution?
The AgCI precipitate would be more desirable than with
All the tools & learning materials you need for study success - in one app.
Get started for free
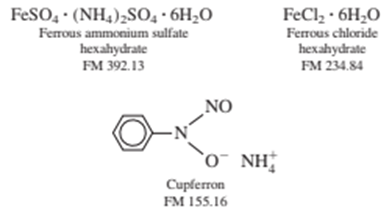
A solid mixture weighing 0.5485 gcontained only ferrous ammonium sulfate hexahydrate and ferrous chloride hexahydrate. The sample was dissolved in , oxidized to with , and precipitated with cupferron. The ferric cupferron complex was ignited to produce 0.1678 gof ferric oxide, (FM 159.69). Calculate the weight percent of Clin the original sample.
Determination of sulfur by combustion analysis produces a mixture of and that can be passed through to convert both into , which is titrated with standard base. When 6.123m g of a substance were burned, the required 3.01mL of0.010576MNaOHfor titration. Find wt% sulfur in the sample.
To find the Ce4+ content of a solid, 4.37 g were dissolved and treated with excess iodate to precipitate Ce(IO3)4. The precipitate was collected, washed well, dried, and ignited to produce 0.104 g of CeO2 (FM 172.114). What was the weight percent of Ce in the original solid?
1.475-g sample containing (FM 138.21), and inert ingredients was dissolved to give 0.100 L of solution. A 25.0-mL aliquot was acidified and treated with excess sodium tetraphenylborate,, to precipitateand
ions completely:
The resulting precipitate amounted to 0.617 g. A fresh 50.0-mL aliquot of the original solution was made alkaline and heated to drive off all the
It was then acidified and treated with sodium tetraphenylborate to give 0.554 g of precipitate. Find the weight percent ofandin the original solid.
Why is sample dropped into the preheated furnace before the oxygen concentration reaches its peak in Figure 27-9?
What do you think about this solution?
We value your feedback to improve our textbook solutions.