Chapter 24: Q11P (page 663)
What is the purpose of derivatization in chromatography? Give an example.
Short Answer
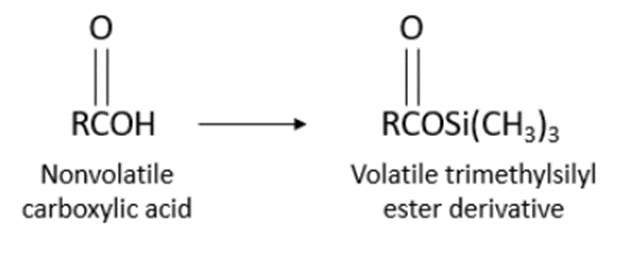
Non-volatile carboxylic acid is converted to volatile trimethylsilyl ester derivative in the process of derivatization.
Learning Materials
Features
Discover
Chapter 24: Q11P (page 663)
What is the purpose of derivatization in chromatography? Give an example.
Non-volatile carboxylic acid is converted to volatile trimethylsilyl ester derivative in the process of derivatization.

All the tools & learning materials you need for study success - in one app.
Get started for free
Oxalate is a naturally occurring substance found in plant foods such as fruits and vegetables. Within the body it can combine with calcium to form kidney or urinary stones. Determination of oxalate in food is important because of the potential for harmful effects on health. Search the literature for a headspace gas chromatography method for the determination of oxalate in food and answer the following questions.
(a) Give the citation (authors, title, journal name, year, volume, pages) for a research paper describing this analysis.
(b) What type of gas chromatography detector is used? Why is this detector appropriate?
(c) What are the precision, limit of quantification, and linear range of the method?
(d) How was the gas chromatographic determination validated?
(e) What alternative methods could be used for determining oxalate in food?
(f) Challenge question: What type of column was used?
Describe how retention time of butanol on a poly (ethylene glycol) column will change with increasing temperature. Use the retention time for butanol in Figure 24-9b as the starting point.
(a) Find the limit of the square-root term as
(unretained solute) and as (infinitely retained solute).
(b) If the column radius is 0.10mmfindfor the two cases in (a).
(c) What is the maximum number of theoretical plates in a 50 -m-long column with a 0.10-mm radius if k=5.0 ?
(d) The phase ratio is defined as the volume of the mobile phase divided by the volume of the stationary phase Derive the relationship between and the thickness of the stationary phase in a wall-coated column and the inside radius of the column
(e) Find kif K=1000,, and r=0.10 mm.
(a) What are the advantages and disadvantages of temperature programming in gas chromatography?
(b) What is the advantage of pressure programming?
(a) How can you improve the resolution between two closely spaced peaks in gas chromatography?
(b) What approach from (a) would be most cost effective (not involve a purchase)?
What do you think about this solution?
We value your feedback to improve our textbook solutions.