Chapter 24: Q21P (page 663)
Describe how retention time of butanol on a poly (ethylene glycol) column will change with increasing temperature. Use the retention time for butanol in Figure 24-9b as the starting point.
Short Answer
NOT GIVEN
Learning Materials
Features
Discover
Chapter 24: Q21P (page 663)
Describe how retention time of butanol on a poly (ethylene glycol) column will change with increasing temperature. Use the retention time for butanol in Figure 24-9b as the starting point.
NOT GIVEN
All the tools & learning materials you need for study success - in one app.
Get started for free
(a) What are the characteristics of an ideal carrier gas?
(b) Why do and He allow more rapid linear velocities in gas chromatography than does, without loss of column efficiency (Figure 24-11)?
The antitumor drug gimatecan is available as nearly pure (S)-enantiomer. Neither pure (R)-enantiomer nor a racemic (equal) mixture of the two enantiomers is available. To measure small quantities of (R)-enantiomer in nearly pure (S)-gimatecan, a preparation was subjected to normal-phase chromatography on each of the enantiomers of a commercial, chiral stationary phase designated (S,S)- and (R,R)-DACH-DNB. Chromatography on the (R,R)-stationary phase gave a slightly asymmetric peak at tr 5 6.10 min with retention factor k 5 1.22. Chromatography on the (S,S)- stationary phase gave a slightly asymmetric peak at tr 5 6.96 min with k 5 1.50. With the (S,S) stationary phase, a small peak with 0.03% of the area of the main peak was observed at 6.10 min.
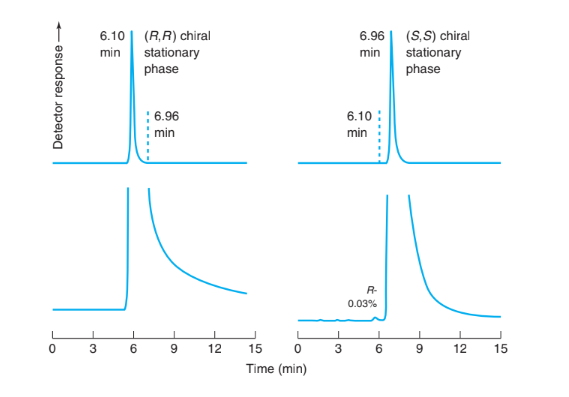
Chromatography of gimatecan on each enantiomer of a chiral stationary phase. Lower traces have enlarged vertical scale. [Data from E. Badaloni, W. Cabri, A. Ciogli, R. Deias, F. Gasparrini, F. Giorgi, A. Vigevani, and C. Villani, “Combination of HPLC ‘Inverted Chirality Columns Approach’ and MS/MS Detection for Extreme Enantiomeric Excess Determination Even in Absence of Reference Samples.” Anal. Chem. 2007, 79, 6013.]
(a) Explain the appearance of the upper chromatograms. Dashed lines are position markers, not part of the chromatogram. What Problems 709 would the chromatogram of pure (R)-gimatecan look like on the same two stationary phases?
(b) Explain the appearance of the two lower chromatograms and why it can be concluded that the gimatecan contained 0.03% of the (R)-enantiomer. Why is the (R)-enantiomer not observed with the (R,R)-stationary phase?
(c) Find the relative retention (a) for the two enantiomers on the (S,S)-stationary phase.
(d) The column provides N 5 6 800 plates. What would be the resolution between the two equal peaks in a racemic (equal) mixture of (R)- and (S)-gimatecan? If the peaks were symmetric, does this resolution provide baseline separation in which signal returns to baseline before the next peak begins?
Explain what is displayed in a reconstructed total ion chromatogram, in selected ion monitoring, and in selected reaction monitoring. Which technique is most selective and which is least selective and why?
Genotoxic compounds damage DNA and cause mutations or cancer.Regulatory guidelines limit genotoxic impurities in pharmaceuticals. Mesityl oxide (4-methyl- 3-penten-2-one) is an intermediate product in the synthesis of some drugs. Search the literature for a gas chromatography method for the determination of mesityl oxide in the pharmaceutical enalapril maleate and answer the following questions.
(a) Give the citation (authors, title, journal name, year, volume, pages) for a research paper describing this analysis.
(b) What is the instrumental method used?
(c) What type of column was used?
(d) What quantification method is used?
(e) What are the precision, limit of detection, limit of quantification, and linear range of the method?
(f) For how long could a standard solution be used?
Efficiency of solid-phase microextraction. Equationgives the mass of analyte extracted into a solid-phase microextraction fiber as a function of the partition coefficient between the fiber coating and the solution.
(a) A commercial fiber with acoating has a film volume of. Suppose that the initial concentration of analyte in solution is
.Use a spreadsheet to prepare a graph showing the mass of analyte extracted into the fiber as a function of solution volume for partition coefficients of and. Let the solution volume vary from .
(b) Evaluate the limit of Equation gets big relative to . Does the extracted mass in your graph approach this limit?
(c) What percentage of the analyte fromof solution is extracted into the fiber when and when
What do you think about this solution?
We value your feedback to improve our textbook solutions.