Chapter 24: Q39P (page 666)
Genotoxic compounds damage DNA and cause mutations or cancer.Regulatory guidelines limit genotoxic impurities in pharmaceuticals. Mesityl oxide (4-methyl- 3-penten-2-one) is an intermediate product in the synthesis of some drugs. Search the literature for a gas chromatography method for the determination of mesityl oxide in the pharmaceutical enalapril maleate and answer the following questions.
(a) Give the citation (authors, title, journal name, year, volume, pages) for a research paper describing this analysis.
(b) What is the instrumental method used?
(c) What type of column was used?
(d) What quantification method is used?
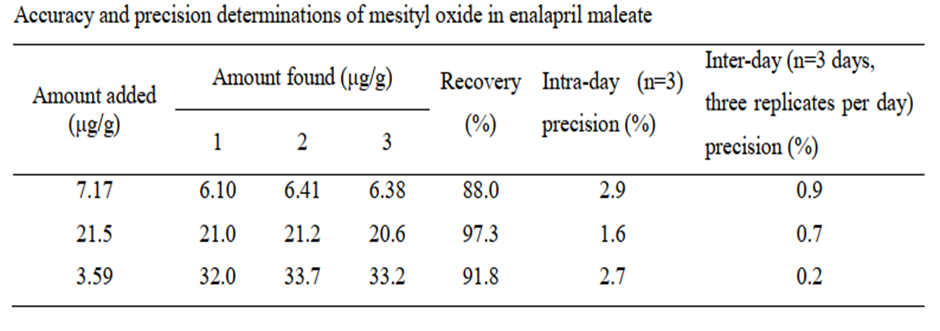
(e) What are the precision, limit of detection, limit of quantification, and linear range of the method?
(f) For how long could a standard solution be used?
Short Answer
(a)Citation:
Authors: Ming Peng, Hongliang Wen, Jian Le, and Yongjian Yang
Title:Trace-level analysis of mesityl oxide in enalapril maleate by gas chromatography with electron ionization mass spectrometry (GC-EI-MS)
Journal name: Analytical Methods
Year: 2012
Volume: 4(12)
Pages: 4063-4065
(b)The instrumental method used here was electron ionization/selective ion monitoring (EI/SIM) method.
(c) An HP 1 ms fused silica capillary column was used.
(d) The international standard curve method was used as the quantification method of mesityl oxide in sample solution and in recovery solution.
(e) This method has an excellent measurement precision (relative standard deviation = 0.9%) and accuracy (relative difference≤2.29%).
The limit of detection in this method is 0.5 µg/g.
The limit of quantitation in this method is 1.8 µg/g.
The quantification of mesityl oxide was linear in the concentration range from 0.18 to 7.17 µg/mL, corresponding to 1.8 ~ 72 µg/g in enalapril maleate.
(f) A standard solution can be used for 24 hr.