Chapter 17: Q39P (page 431)
What are the advantages of using a microelectrode for
voltametric measurements?

Short Answer
The benefits of microelectrodes in voltammetry techniques has been explained.
Learning Materials
Features
Discover
Chapter 17: Q39P (page 431)
What are the advantages of using a microelectrode for
voltametric measurements?

The benefits of microelectrodes in voltammetry techniques has been explained.
All the tools & learning materials you need for study success - in one app.
Get started for free
In a coulometric Karl Fischer water analysis, 25.00 mL of pure "dry" methanol required 4.23 C to generate enough I2 to react with residual H2O in the methanol. A suspension of 0.8476 g of finely ground polymeric material in 25.00 mL of the same "dry" methanol required 63.16 C. Find the wt %H2O in the polymer.
The chlor-alkali process 54in which seawater is electrolyzed to make CI2 andNaOH, is the second most important commercial electrolysis, behindAIproduction.
Anode:
HgCathode:
The Nafion membrane (page 421) used to separate the anode and cathode compartments resists chemical attack. Its anionic side chains permit conduction ofNa+ , but not anions. The cathode compartment contains pure water, and the anode compartment contains seawater from which Ca2+ and Mg2+ have been removed. Explain how the membrane allows NaOH to be formed free ofNaCI.
Aunknown containing (leadLactate, FM 385.3) plus inert material was electrolyzed to produce . Was the PbO2deposited at the anode or at the cathode? Find the weight percent of lead lactate in theUnknown.
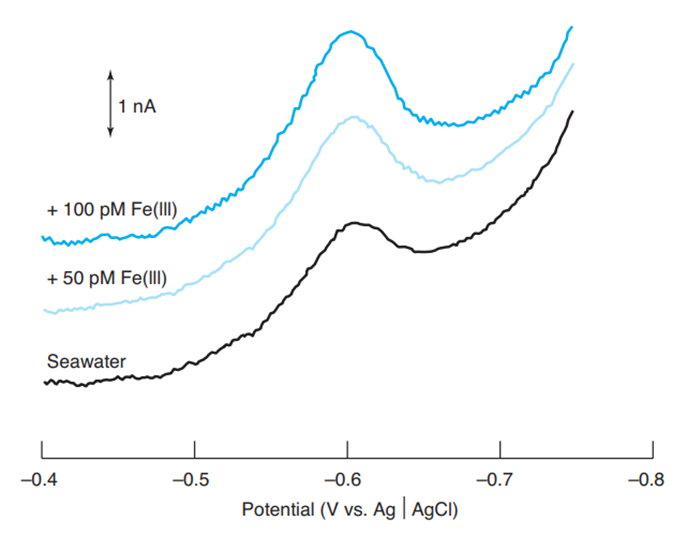
From the two standard additions of 50 pm Fe(III) in the figure, find the concentration of Fe(III) in the seawater. Estimate where the baseline should be drawn for each trace and measure the peak height from the baseline. Consider the volume to be constant for all three solutions.
The figure shows the behavior of Ptand Ag cathodes at whichreduction oftooccurs. Explain why the two curves arenot superimposed.
Current versus voltage forPtand Agelectrodes in O2 -free, aqueous H2SO4
Adjusted topH3.2.
What do you think about this solution?
We value your feedback to improve our textbook solutions.