Chemical oxygen demand by coulonetry. An electrochemical device incorporating photooxidation on a \({\rm{Ti}}{{\rm{O}}_2}\) surface could replace refluxing with \({{\rm{C}}_2}{\rm{O}}_7^{2 - }\) to measure chemical oxygen demand (Box 16-2). The diagram shows a working electrode beld at \( + 0.30\;{\rm{V}}\) versus \({\rm{Ag}}\mid {\rm{AgCl}}\) and coated with nanoparticles of 'TiO . Wltraviolet2inradiation generates electrons and holes in \({{\rm{T}}_1}{{\rm{O}}_2}\). Holes oxidize
organic matter at the surface. Electrons reduce \({{\rm{H}}_2}{\rm{O}}\) at the auxiliary electrode in a compartment connected to the working compartment by a salt bridge. The sample compartment is only 0.18 mm thick with a volume of \(13.5\mu \,{\rm{L}}\). It requires \(\~1\;\,{\rm{min}}\) for all organic matter to diffuse to the \({\rm{Ti}}{{\rm{O}}_2}\) surface and be exhaustively oxidized.
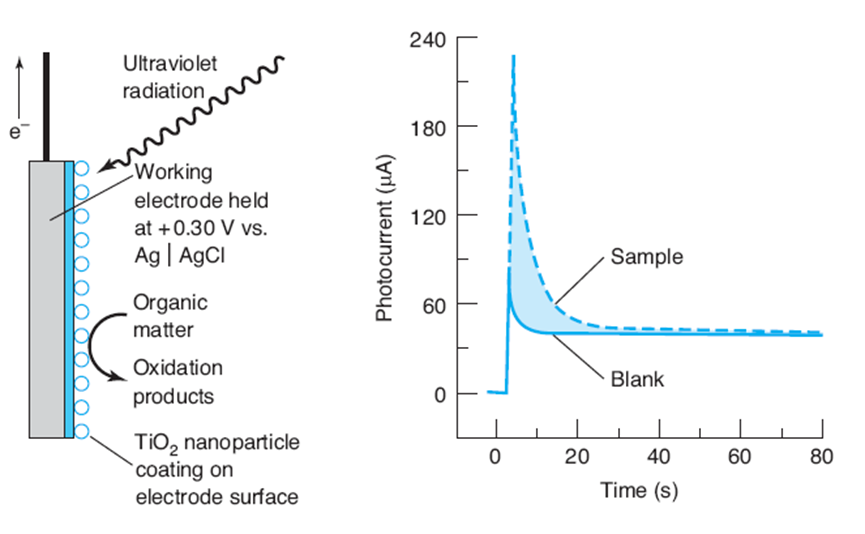
Left: Working electrode. Fight Photocument response for sample and blank Both solutions contain \(2{\rm{M}}\,{\rm{NaNO}}\). (Dst from H zhso, D. fisng. 5 . zhang K. Cutteral, and R. Jshn, "Development of a Drect Fhotselectrocherrical Method for Deterrination of Gherrical Ouygen Demand," And. Chan. 2004, 76 155.)
The blank curve in the graph shows the response when the sample compartment contains just electrolyte. Before inradiation, no current is observed. Ultraviolet radiation causes a spike in the current, followed by a decrease to a steady level near \(40\mu \). This current arises from oxidation of water at the \({\rm{Ti}}{{\rm{O}}_2}\)sufface under ultraviolet exposure. The upper curve sbows the same experiment, but with wastewater in the sample compartment. The increased current arises from oxidation of organic matter. When the organic matter is consumed, the cument decreases to the blank level. The area between the two curves tells us how many electrons flow from oxidation of organic matter in the sample.
- Balance the oxidation half-reaction that occurs in this cell:
\({{\rm{C}}_e}{{\rm{H}}_k}{{\rm{O}}_a}\;{{\rm{N}}_s}{{\rm{X}}_x} + {\rm{A}}{{\rm{H}}_2}{\rm{O}} \to {\rm{BC}}{{\rm{O}}_2} + {\rm{CX}} + {\rm{DN}}{{\rm{H}}_3} + {\rm{E}}{{\rm{H}}^ + } + {\rm{F}}{{\rm{e}}^ - }\)
where X is any halogen. Express the stoichiometry coefficients A, B, C, D, E, and F in terms of c, h, o, n, and x.
- How many molecules of \({{\rm{O}}_2}\)are required to balance the halfreaction in part (a) by reduction of oxygen (\({{\rm{O}}_2} + 4{{\rm{H}}^ + } + 4{{\rm{e}}^ - } \to 2{{\rm{H}}_2}{\rm{O}}\))?
- The area between the two curves in the graph is \(\int_0^\infty {({I_{{\rm{sample }}}}} - {I_{blank}})dt = 9.43\,{\rm{mC}}{\rm{.}}\) This is the number of electrons liberated by complete oxidation of the sample. How many moles of \({{\rm{O}}_2}\) would be required for the same oxidation?
- Chemical oxygen demand (COD) is expressed as mg of \({{\rm{O}}_2}\) required to oxidize 1 L of sample. Find the COD for this sample.
- If the only caidizable substance in the sample were \({{\rm{C}}_9}{{\rm{H}}_6}{\rm{N}}{{\rm{O}}_2}{\rm{CIB}}{{\rm{r}}_2}\). what is its concentration in molL?




