Chapter 17: Q43P (page 431)
Explain how the endpoint is detected in a Karl Fischer titration in Figure 17-35.
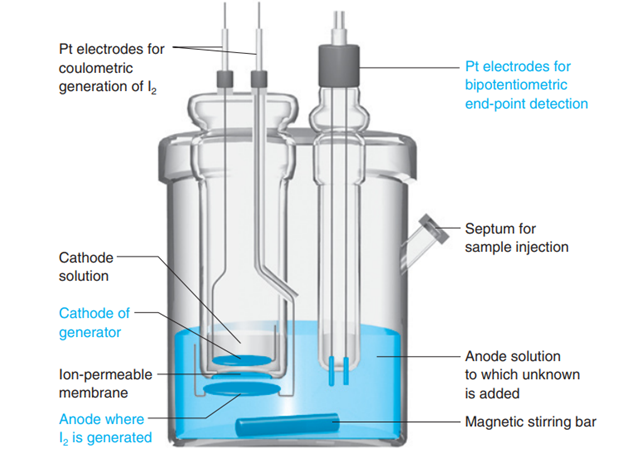
Short Answer
The process of detection of endpoint in Karl Fischer titration.
Learning Materials
Features
Discover
Chapter 17: Q43P (page 431)
Explain how the endpoint is detected in a Karl Fischer titration in Figure 17-35.

The process of detection of endpoint in Karl Fischer titration.
All the tools & learning materials you need for study success - in one app.
Get started for free
17-18. can be analysed by titration with coulometric ally generated
role="math" localid="1654768790950"
To of sample were added 4gof KI. Electrolysis required812s at. 52.6mA . Calculate the concentration ofrole="math" localid="1654769975824" in the sample.
Fundamentals of Electrolysis
17-9. A lead-acid battery in a car has six cells in series, each delivering close to2.0Vfor a total of 12V when the battery is discharging. Recharging requires per cell, or for the entire battery. 55Explain these observations in terms of Equation 17-6.
How many hours are required for 0.100 mol of electrons to flow through a circuit if the current is 1.00 A?
Chlorine has been used for decades to disinfect drinking water. An undesirable side effect of this treatment is reaction with organic impurities to create organochlorine compounds, some of which could be toxic. Monitoring total organic halide (designated TOX) is required for many water providers. A standard procedure for TOX is to pass water through activated charcoal, which adsorbs organic compounds. Then the charcoal is combusted to liberate hydrogen halides:
HX is absorbed into aqueous solution and measured by coulometric titration with a silver anode:
When 1.00 L of drinking water was analyzed, a current of 4.23 mA was required for 387 s. A blank prepared by oxidizing charcoal required 6 s at 4.23 mA. Express the TOX of the drinking water as mol halogen/L. If all halogen is chlorine, express the TOX as .
The figure shows the behavior of Ptand Ag cathodes at whichreduction oftooccurs. Explain why the two curves arenot superimposed.
Current versus voltage forPtand Agelectrodes in O2 -free, aqueous H2SO4
Adjusted topH3.2.
What do you think about this solution?
We value your feedback to improve our textbook solutions.