Chapter 12: Q8P (page 283)
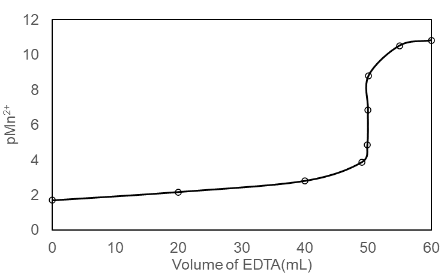
Consider the titration of 25.0 mL of 0.020 0 M MnSO4 with 0.010 0 M EDTA in a solution buffered to pH 8.00. Calculate pMn2+ at the following volumes of added EDTA and sketch the titration curve:
(a) 0 mL (b) 20.0 mL (c) 40.0 mL (d) 49.0 mL (e) 49.9 mL (f) 50.0 mL (g) 50.1 mL
(h) 55.0 mL (i) 60.0 mL
Short Answer
(a) For 0 mL the value of pMn2+is 1.7.
(b) For 20 mL the value ofpMn2+is 2.175.
(c) For 40 mL the value ofpMn2+is 2.81.
(d) For 49 mL the value ofpMn2+is 3.87.
(e) For 49.9 mL the value ofpMn2+is 4.87.
(f) For 50 mL the value of pMn2+is 6.85.
(g) For 50.1 mL the value of pMn2+is 8.69.
(h) For 55 mL the value of pMn2+is 10.52.
(i) For 60 mL the value of pMn2+is 10.82.