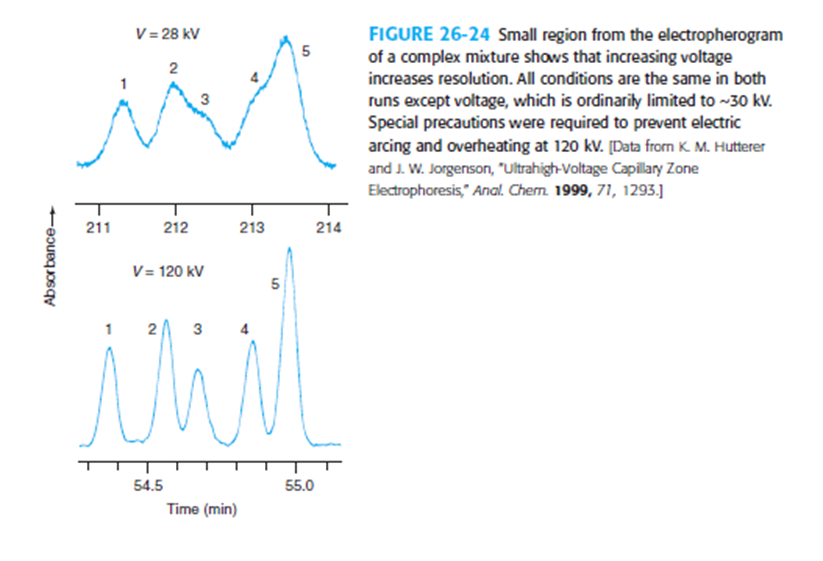
Chapter 26: Q28P (page 748)
Electroosmotic velocities of buffered solutions are shown for a bare silica capillary and one with aminopropyl groups (silica—SiOCH2CH2CH2NH2) covalently attached to the wall. A positive sign means that flow is toward the cathode. Explain the signs and relative magnitudes of the velocities.

Short Answer
Negative wall - flow towards the cathode.
Positive wall - flow towards the anode.
→ the protonation of silanol and amines has an effect on the velocity.