Chapter 6: Q19P (page 142)
A solution contains and . Can 99%of be precipitated by sulphate without precipitating? What will be the concentration ofwhenbegins to precipitate?
Short Answer
97 % of was precipitated.
Learning Materials
Features
Discover
Chapter 6: Q19P (page 142)
A solution contains and . Can 99%of be precipitated by sulphate without precipitating? What will be the concentration ofwhenbegins to precipitate?
97 % of was precipitated.
All the tools & learning materials you need for study success - in one app.
Get started for free
(a) Using only from Table 6-3, calculate how many moles
of will dissolve in 1.00 L of water.
(b) How will the solubility calculated in part (a) be affected by the reaction in Table 6-3?
For the reaction , the concentrations at equilibrium are found to be
A:
B:
C:
D:
E:
Find the numerical value of the equilibrium constant that would appear in a conventional table of equilibrium constants.
Question: (a) From Kw in Table 6-1, calculate the pH of pure water at 00,200, and 400C.
(b) For the reaction , at 250C. In this equation, D stands for deuterium, which is the isotope 2H. What is the pD (=-log[D+]) for neutral D2O?
Write the expression for the equilibrium constant for each of the following reactions. Write the pressure of a gas, X , as .
(a)
(b)
Question: Histidine is a triprotic amino acid:
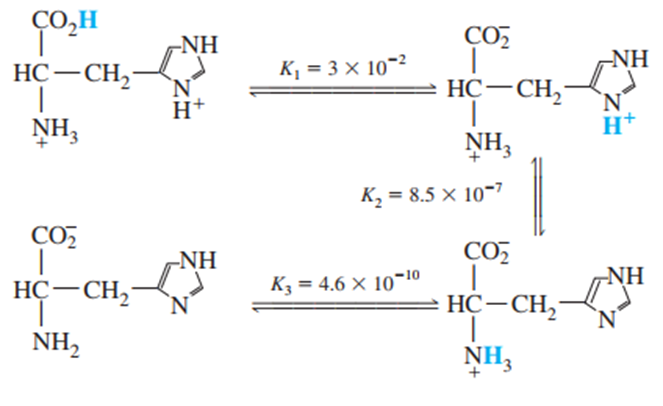
Find the value of the equilibrium constant for the reaction
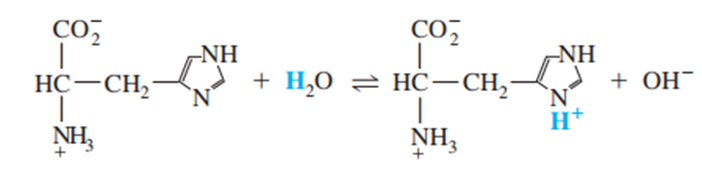
What do you think about this solution?
We value your feedback to improve our textbook solutions.