Chapter 19: Q3P (page 484)
When are isosbestic points observed and why?
Short Answer
When spectra of two compounds with a constant total concentration cross at any wavelength, the isosbestic points are observed.
Learning Materials
Features
Discover
Chapter 19: Q3P (page 484)
When are isosbestic points observed and why?
When spectra of two compounds with a constant total concentration cross at any wavelength, the isosbestic points are observed.
All the tools & learning materials you need for study success - in one app.
Get started for free
The spreadsheet lists molar absorptivities of three dyes and the absorbance of a mixture of the dyes in a 1.000-cm cell. Use the least-squares procedure in Figure 19-3 to find the concentration of each dye in the mixture.
Iodine reacts with mesitylene to form a complex with an absorption maximum at 332 nm in CCl4 solution:
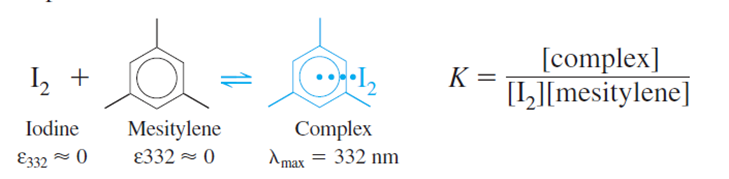
(b)Spectrophotometric data for this reaction are shown in the table.Becausewe can say that [mesitylene][mesitylene]tot. Prepare a graph ofversusand find the equilibrium constant and molar absorptivity of the complex.

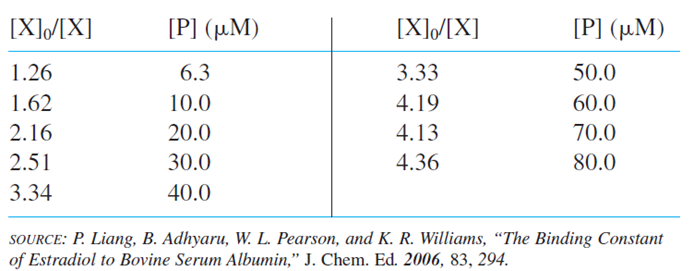
Scatchard plot for binding of estradiol to albumin. Data in the table come from a student experiment to measure the binding constant of the radioactively labeled hormone estradiol (X)to the protein, bovine serum albumin was equilibrated with various concentrations of albumin for A small fraction of unbound estradiol was removed by solid phase microextraction () and measured by liquid scintillation counting. Albumin is present in large excess, so its concentration in any given solution is essentially equal to its initial concentration in that solution. Call the initial concentration of estradiol and the final concentration of unbound estradiol [X]. Then bound estradiol isand the equilibrium constant is
which you can rearrange to
localid="1663648487221"
A graph of versus [P]should be a straight line with a slope of K.The quotient is equal to the counts of radioactive estradiol extracted from a solution without albumin divided by the counts of estradiol extracted from a solution with estradiol. (b) What fraction of estradiol is bound to albumin at the first and last points?
Two ways to analyze a mixture. Figure 19-5 shows the spectrum of the indicator bromothymol blue adjusted to several pH values. The spectrum at pHis that of the pure blue form and the spectrum at pH 1.8is that of the pure yellow form. At other pHvalues, there is a mixture of the two forms. The total concentration isand the path length isin all spectra. For the purpose of calculation, assume that there are more than two significant digits in concentration and path length. Absorbance at the dots on three of the curves in Figure 19-5 is given in the table.
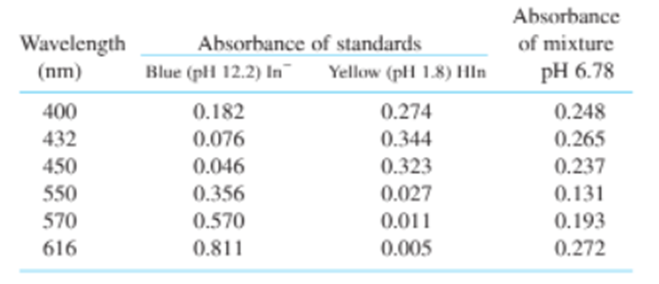
(a) Prepare a spreadsheet like Figure 19-3 to use absorption at all six wavelengths to findin the mixture. Comment on the sum.
(b) Fromin the mixture, and fromfor HIn,calculate theof the mixture. (This calculation is the source of pH labels in the figure.)
(c) Use Equations 19-6 at the peak wavelengths ofto findin the mixture. Compare your answers to those in (a). Which answers, (a) or (c), are probably more accurate? Why?
Chemical equilibrium and analysis of a mixture. (Warning! This is a long problem.) A remote optical sensor for in the ocean was designed to operate without the need for calibration.33
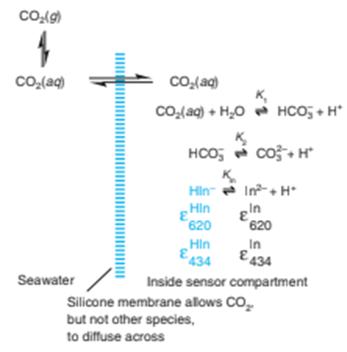
The sensor compartment is separated from seawater by a silicone membrane through which , but not dissolved ions, can diffuse. Inside the sensor, equilibrates with and . For each
measurement, the sensor is flushed with fresh solution containingbromothymol blue indicator. All indicator is in the formnear neutral pH, so we can
write two mass balances:
has an absorbance maximum at 434 nm andhas a maximum at 620 nm. The sensor measures the absorbance ratio reproducibly without need for calibration. From this ratio, we can findin the seawater as outlined here:
(a).From Beer’s law for the mixture, write equations forin terms of the absorbance at 620 and 434 nmThen show that
(A)
(b) From the mass balance (1) and the acid dissociation constant
, show that
(B)
(C)
(c) Show that (D)
(d) From the carbonic acid dissociation equilibria, show that
(e) Write the charge balance for the solution in the sensor compartment. Substitute in expressions B, C, E, and F forHln,
(f) Suppose that the various constants have the following values:
From the measured absorbance ratio=2.84, findin the seawater.
(g) Approximately what is the ionic strength inside the sensor compartment? Were we justified in neglecting activity coefficients in working this problem?
What do you think about this solution?
We value your feedback to improve our textbook solutions.