Chapter 19: Q1TY (page 464)
Find the concentration of [X] if the absorbance are 0.700 at 272 nm and
0.550 at 327 nm.
Short Answer
The concentration of [X] if the absorbance are 0.700 at 272 nm and
0.550 at 327 nm is .
Learning Materials
Features
Discover
Chapter 19: Q1TY (page 464)
Find the concentration of [X] if the absorbance are 0.700 at 272 nm and
0.550 at 327 nm.
The concentration of [X] if the absorbance are 0.700 at 272 nm and
0.550 at 327 nm is .
All the tools & learning materials you need for study success - in one app.
Get started for free
What is the advantage of a time-resolved emission measurement with Eu3+versus measurement of fluorescence from organic chromophores?
This problem can be worked with Equations 19-6 on a calculator or with the spreadsheet in Figure 19-4. Transferrin is the iron-transport protein found in blood. It has a molecular mass of 81 000 and carries twoions. Desferrioxamine B is a chelator used to treat patients with iron overload (see the opening of Chapter 12). It has a molecular mass of about 650 and can bind oneFe31. Desferrioxamine can take iron from many sites within the body and is excreted (with its iron) through the kidneys. Molar absorptivities of these compounds (saturated with iron) at two wavelengths are given in the table. Both compounds are colorless (no visible absorption) in the absence of iron.
(a) A solution of transferrin exhibits an absorbance of 0.463 at 470 nm in a 1.000-cm cell. Calculate the concentration of transferrin in milligrams per milliliter and the concentration of bound iron in micrograms per milliliter.
(b) After adding desferrioxamine (which dilutes the sample), the absorbance at 470 nm was 0.424, and the absorbance at 428 nm was 0.401. Calculate the fraction of iron in transferrin and the fraction in desferrioxamine. Remember that transferrin binds two iron atoms and desferrioxamine binds only one.
The spreadsheet lists molar absorptivities of three dyes and the absorbance of a mixture of the dyes in a 1.000-cm cell. Use the least-squares procedure in Figure 19-3 to find the concentration of each dye in the mixture.
Iodine reacts with mesitylene to form a complex with an absorption maximum at 332 nm in CCl4 solution:
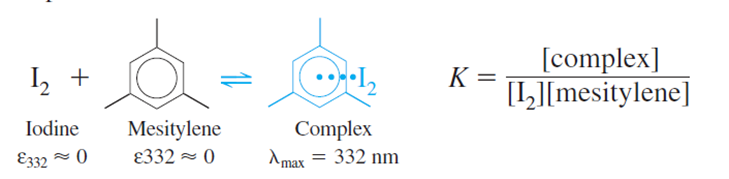
(b)Spectrophotometric data for this reaction are shown in the table.Becausewe can say that [mesitylene][mesitylene]tot. Prepare a graph ofversusand find the equilibrium constant and molar absorptivity of the complex.

This problem can be worked by calculator or with the spreadsheet in Figure 19-4. Consider compounds X and Y in the example labeled “Analysis of a Mixture, Using Equations 19-6” on page 464. Find [X] and [Y] in a solution whose absorbance is 0.233 at 272 nm and 0.200 at 327 nm in a 0.100-cm cell.
What do you think about this solution?
We value your feedback to improve our textbook solutions.