Chapter 11: Q35P (page 261)
Prepare a second derivative graph to find the end point from the following titration data.
Short Answer
The graph is shown below
Learning Materials
Features
Discover
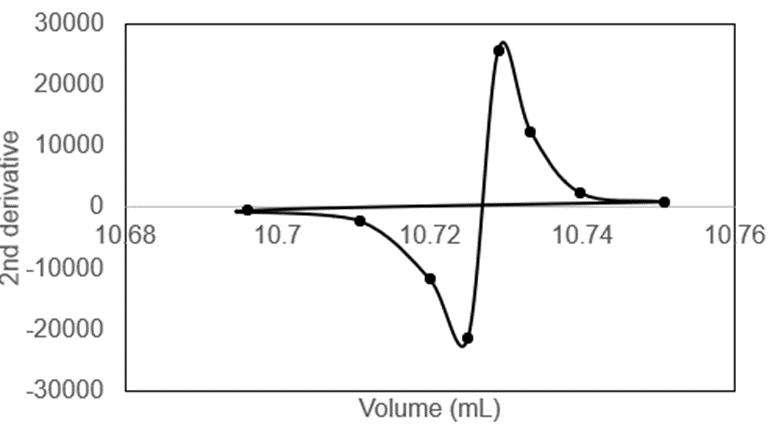
Chapter 11: Q35P (page 261)
Prepare a second derivative graph to find the end point from the following titration data.

The graph is shown below

All the tools & learning materials you need for study success - in one app.
Get started for free
The balance says that you have weighed out 1.023 g of tris tostandardize a solution of HCl. Use the buoyancy correction in Section 2-3 and the density in Table 11-4 to determine how many grams you have really weighed out. The volume of HCl required to react with the tris was 28.37 mL. Does the buoyancy correction introduce a random or a systematic error into the calculated molarity of HCl? What is the magnitude of the error expressed as a percentage? Is the calculated molarity of HCl higher or lower than the true molarity?
Consider the titration of weak acid with . At what fraction of does ? At what fraction of does localid="1655007666473" ? Use these two points, plus to sketch the titration curve for the reaction of of localid="1655007634810" anilinium bromide (aminobenzene. ) withlocalid="1655007889912" .
Question:Effect of concentration in the titration of weak acid with strong base.Use your spreadsheet from Problem 11-66 to prepare a family of titration curves for pKa = 6, with the following combinations of concentrations: (a) Ca = 20 mM, Cb = 100 mM; (b) Ca = 2 mM, Cb = 10 mM; (c) Ca = 0.2 mM, Cb = 1 mM.
Titrating nicotine with strong acid.Prepare a spreadsheet to reproduce the lower curve in Figure 11-4.
A certain acid-base indicator exists in three colored forms:

The units of molar absorptivity, ε, are M-1 cm-1. A solution containing 10.0 mL of 5.00 × 10-4 M indicator was mixed with 90.0 mL of 0.1 M phosphate buffer (pH 7.50). Calculate the absorbance of this solution at 435 nm in a 1.00-cm cell.
What do you think about this solution?
We value your feedback to improve our textbook solutions.