Chapter 11: Q67P c (page 263)
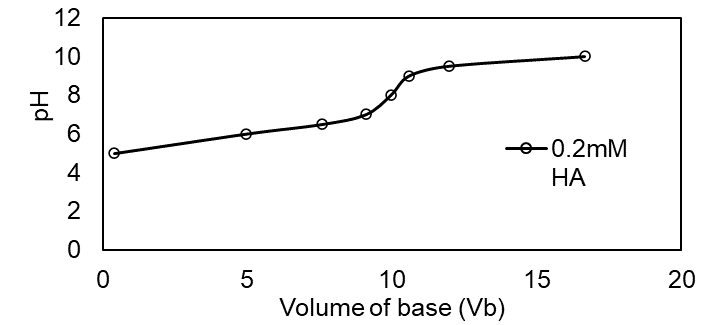
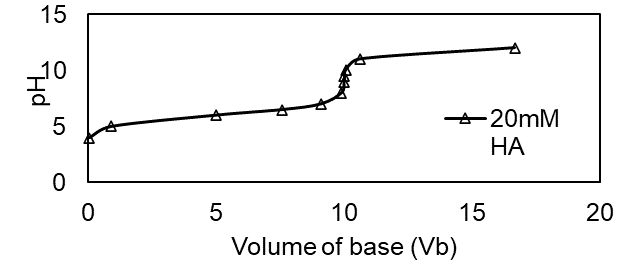
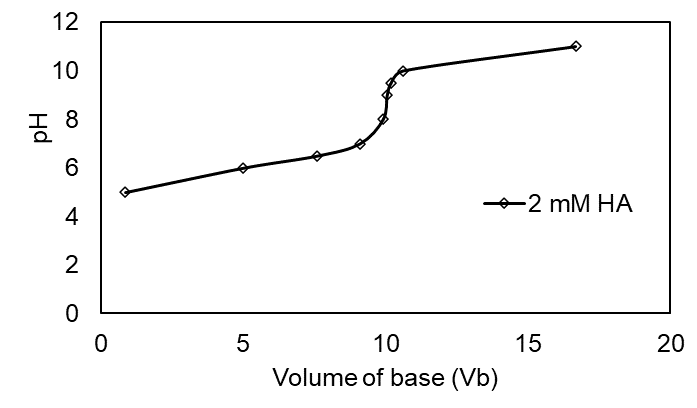
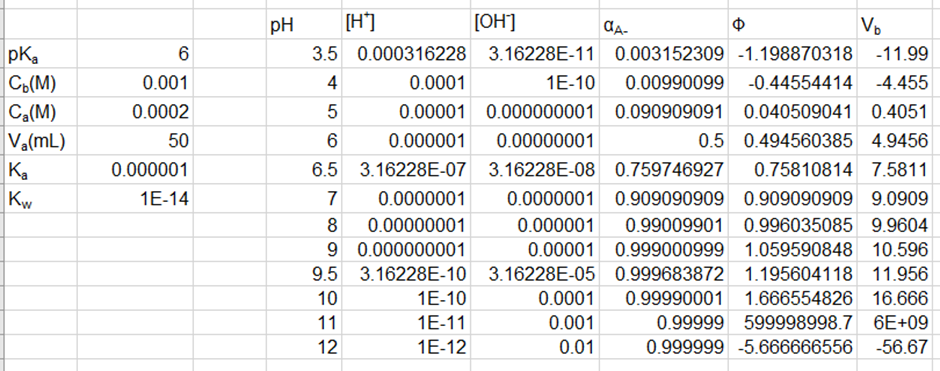
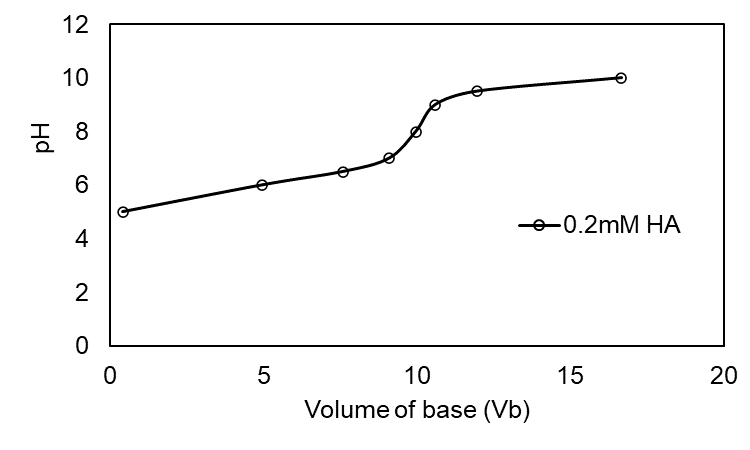
Question:Effect of concentration in the titration of weak acid with strong base.Use your spreadsheet from Problem 11-66 to prepare a family of titration curves for pKa = 6, with the following combinations of concentrations: (a) Ca = 20 mM, Cb = 100 mM; (b) Ca = 2 mM, Cb = 10 mM; (c) Ca = 0.2 mM, Cb = 1 mM.
Short Answer
The plot of the family of titration curves for pKa =6 is shown below
(c)