Chapter 11: Q71P (page 263)
Titrating nicotine with strong acid.Prepare a spreadsheet to reproduce the lower curve in Figure 11-4.
Short Answer
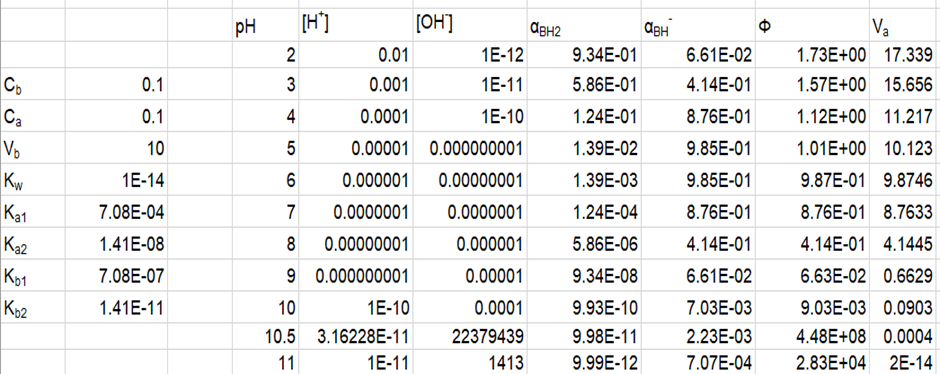
The spreadsheet developed is shown below
Learning Materials
Features
Discover
Chapter 11: Q71P (page 263)
Titrating nicotine with strong acid.Prepare a spreadsheet to reproduce the lower curve in Figure 11-4.
The spreadsheet developed is shown below

All the tools & learning materials you need for study success - in one app.
Get started for free
Titration on Diprotic Systems
11 - 30. This problem deals with the amino acid cysteine, which we will abbreviate .
(a) Asolution was prepared by dissolving dipotassium cysteine, in water. Then 40.0 mLof this solution were titrated with . Calculate the at the first equivalence point.
(b) Calculate the quotient in a solution of 0.0500M cysteinium bromide (the salt).
What is the equilibrium constant for the reaction between benzylamine and?
11-21. The base, whose anion is dibasic, was titrated withHCI to give curveb in Figures 11-4. Is the first equivalence point Hthe isoelectric point or the isoionic point?
Explain why sodium amide (NaNH2) and phenyl lithium (C6H5Li) are levelled to the same base strength in aqueous solution. Write the chemical reactions that occur when these reagents are added to water.
Write the chemical reactions (including structures of reactants and products) that occur when the amino acid histidine is titrated with perchloric acid. (Histidine is a molecule with no net charge.) A solution containing \(25.0\;{\rm{mL}}\) of \(0.0500{\rm{M}}\)histidine was titrated with \(0.0500{\rm{M}}\)\({\rm{HCl}}{{\rm{O}}_4}\). Calculate the pH at the following values of\({V_{\rm{a}}}:0,4.0,12.5,25.0,26.0\)and\(50.0\;{\rm{mL}}\).
What do you think about this solution?
We value your feedback to improve our textbook solutions.