Chapter 25: Q4P (page 1131)
Draw the structure of an optically active fat that, when hydrolyzed under acidic conditions, gives the same products as the fat in Problem 3.
Short Answer
Structure of optically active fat
Learning Materials
Features
Discover
Chapter 25: Q4P (page 1131)
Draw the structure of an optically active fat that, when hydrolyzed under acidic conditions, gives the same products as the fat in Problem 3.

Structure of optically active fat
All the tools & learning materials you need for study success - in one app.
Get started for free
Treating with a strong base such as sodium tert-butoxide followed by the addition of acid converts it to . Propose a mechanism for this reaction
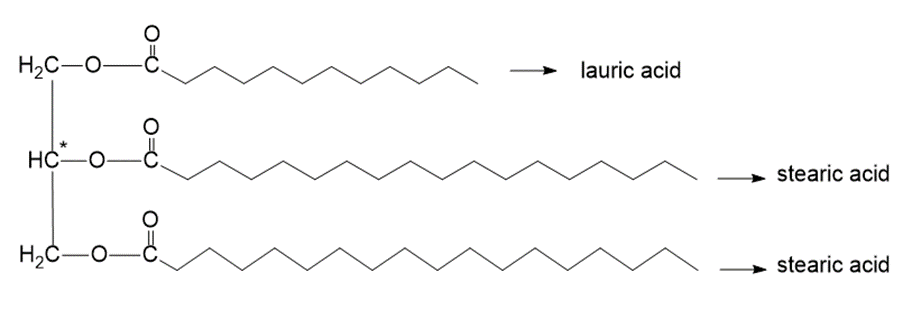
Draw the structure of an optically inactive fat that, when hydrolyzed under acidic conditions, gives glycerol, one equivalent of lauric acid, and two equivalents of stearic acid.
The membrane phospholipids in deer have a higher degree of unsaturation in cells closer to the hoof than in cells closer to the body. Why is this trait important for survival?
Mark the isoprene units in menthol, -selinene, and camphor.
What compounds are obtained when (+)-limonene (its structure is on page 1144) reacts with O3followed by dimethyl sulfide?
What do you think about this solution?
We value your feedback to improve our textbook solutions.