Chapter 25: Q29P (page 1149)
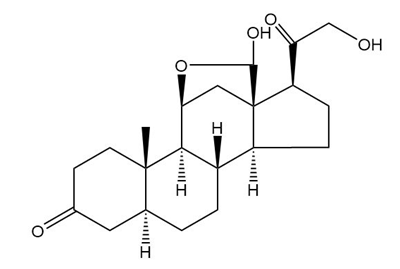
Aldosterone is in equilibrium with its cyclic hemiacetal. Draw the hemiacetal form of aldosterone.
Short Answer
Hemiacetal form of aldosterone:
Learning Materials
Features
Discover
Chapter 25: Q29P (page 1149)
Aldosterone is in equilibrium with its cyclic hemiacetal. Draw the hemiacetal form of aldosterone.
Hemiacetal form of aldosterone:

All the tools & learning materials you need for study success - in one app.
Get started for free
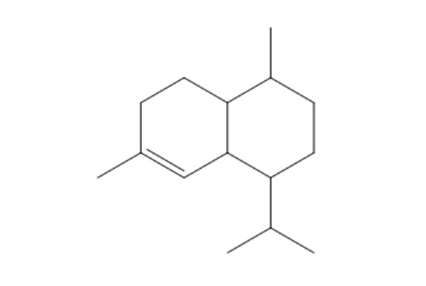
If the following sesquiterpene is synthesized in a medium containing acetate with a 14C-labeled carbonyl carbon, which carbons will be labeled?
Treating with a strong base such as sodium tert-butoxide followed by the addition of acid converts it to . Propose a mechanism for this reaction
A colony of bacteria accustomed to an environment with a temperature of was moved to an identical environment, except that its temperature was . The higher temperature increased the fluidity of the bacterial membranes. How can the bacteria regain their original membrane fluidity?
An optically active monoterpene (compound A) with molecular formula C10H18Oundergoes catalytic hydrogenation to form an optically inactive compound with the molecular formula C10H20O(compound B). When compound B is heated with acid, followed by reaction withand then with O3dimethyl sulfide, one of the products obtained is 4-methyl cyclohexanone. Give possible structures for compounds A and B.
What compounds are obtained when (+)-limonene (its structure is on page 1144) reacts with O3followed by dimethyl sulfide?
What do you think about this solution?
We value your feedback to improve our textbook solutions.