Chapter 25: Q20P (page 1144)
Propose a mechanism for the conversion of E isomer of geranyl pyrophosphate to Z isomer.
Short Answer
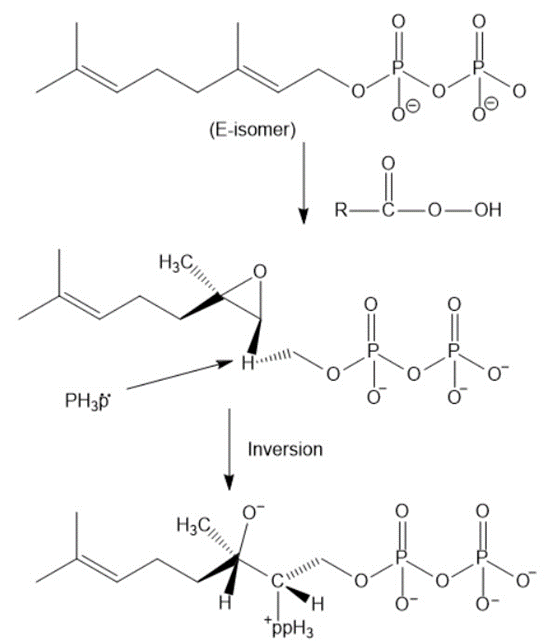
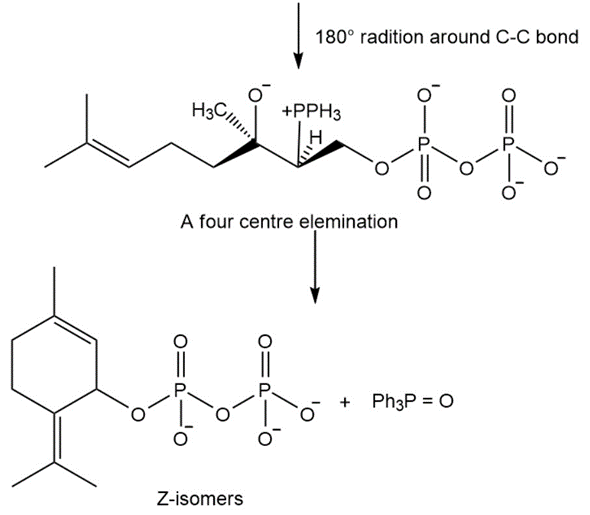
The mechanism for the conversion of E isomer of geranyl pyrophosphate to Z isomer.
Learning Materials
Features
Discover
Chapter 25: Q20P (page 1144)
Propose a mechanism for the conversion of E isomer of geranyl pyrophosphate to Z isomer.
The mechanism for the conversion of E isomer of geranyl pyrophosphate to Z isomer.


All the tools & learning materials you need for study success - in one app.
Get started for free
Membranes contain proteins. Integral membrane proteins extend partly or completely through the membrane, whereas peripheral membrane proteins are found on the inner or outer surface of the membrane. What is the likely difference in the amino acid composition of integral and peripheral membrane proteins?
Treating with a strong base such as sodium tert-butoxide followed by the addition of acid converts it to . Propose a mechanism for this reaction
Do the identities of and in phosphatidic acid affect the configuration of the asymmetric center?
Dipalmitoyl phosphatidylcholine is a surfactant found in the lining of the lungs. It prevents the lungs from collapsing when the lung volume is low andprotects the lungs from injuries caused by inhaled particles. Draw its structure
An optically active fat, when completely hydrolyzed, yields twice as much stearic acid as palmitic acid. Draw the structure of the fat.
What do you think about this solution?
We value your feedback to improve our textbook solutions.