Chapter 18: Q4P (page 877)
Show the mechanism for the generation of the acylium ion if an acid anhydride is used instead of an acyl chloride for the source of the acylium ion.
Short Answer
The answer is,
 Mechanism
Mechanism
Learning Materials
Features
Discover
Chapter 18: Q4P (page 877)
Show the mechanism for the generation of the acylium ion if an acid anhydride is used instead of an acyl chloride for the source of the acylium ion.
The answer is,
 Mechanism
Mechanism
All the tools & learning materials you need for study success - in one app.
Get started for free
What reagents are required to carry out the following synthesis?
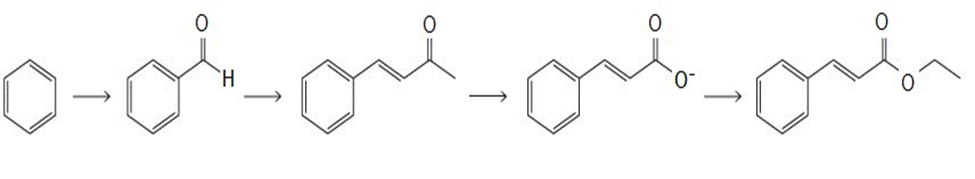
Describe how 3-methyl-1-phenyl-3-pentanol can be prepared from benzene. You can use any inorganic reagents and solvents, and any organic reagentsprovided they contain no more than two carbons.
Question:a. How can aspirin be synthesized from benzene?b. Ibuprofen is the active ingredient in pain relievers such as Advil, Motrin and Nuprin. How can ibuprofen be synthesized from benzene?c. Acetaminophen is the active ingredient in Tylenol. How can acetaminophen be synthesized from benzene?
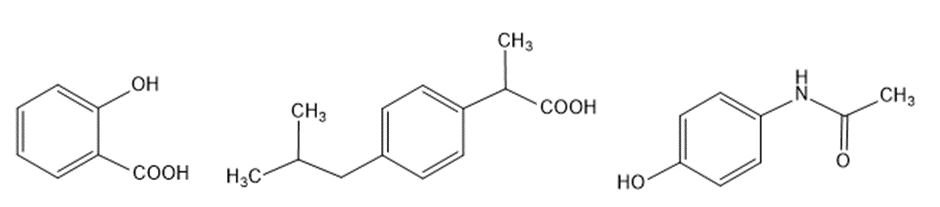
aspirin ibuprofen acetaminophen
Question: Propose a mechanism for each of the following reactions:
a.)
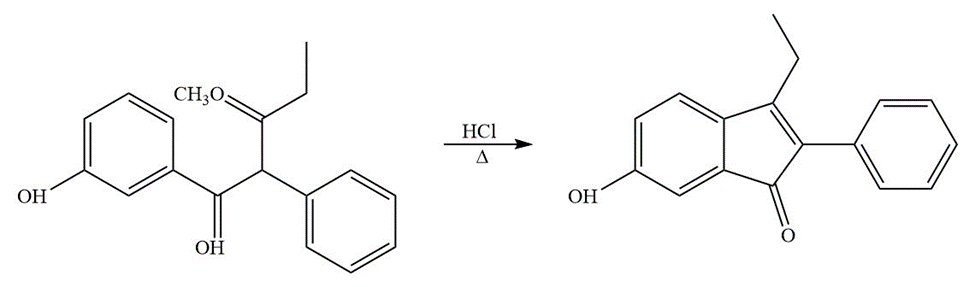
b.)
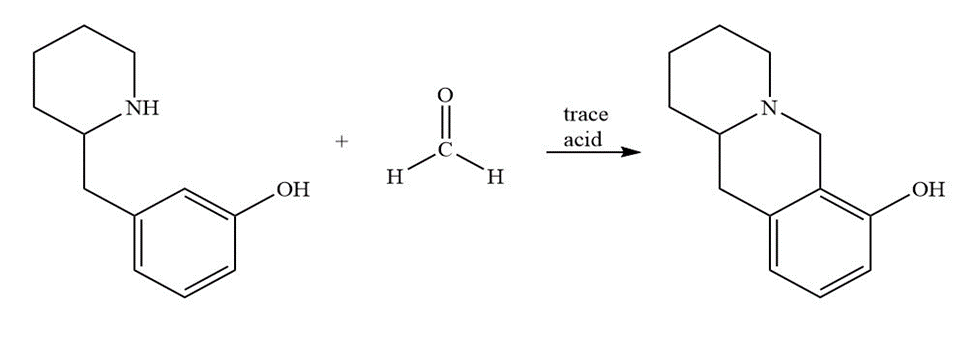
A student had prepared three ethyl-substituted benzaldehydes, but neglected to label them. The student at the next bench said they could be identified by brominating a sample of each and determining how manybromo-substituted products are formed. Is the student’s advice sound?
What do you think about this solution?
We value your feedback to improve our textbook solutions.