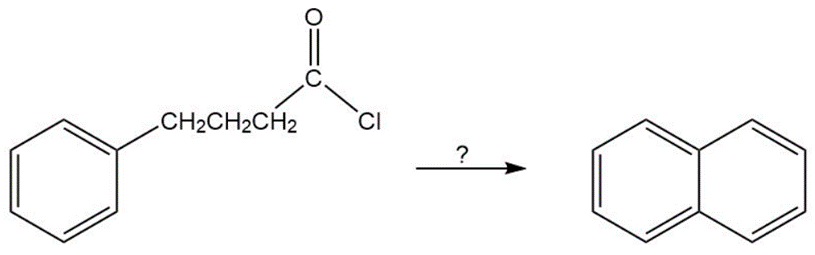
Chapter 18: Q 89 P (page 920)
Question: p-fluoronitrobenzene is more reactive toward hydroxide ion than is p-chloronitrobenzene. What does this tell you about the rate-determining step fornucleophilic aromatic substitution?
Short Answer
The rate-determining step in a nucleophilic aromatic substitution reaction is the slow step addition of hydroxide ion. The electronegativity difference between chlorine and fluorine is the reason behind the addition of hydroxide ion to p-Fluoronitrobenzene.


 Iidocain
Iidocain