Chapter 18: Q-18-2P (page 873)
Question: Why does hydration inactivates FeBr3 ?
Short Answer
The hydrated ferric bromide accepted a lone pair of electrons from the water, and they cannot accept more lone pair electrons from other atoms.
Learning Materials
Features
Discover
Chapter 18: Q-18-2P (page 873)
Question: Why does hydration inactivates FeBr3 ?
The hydrated ferric bromide accepted a lone pair of electrons from the water, and they cannot accept more lone pair electrons from other atoms.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:a. How can aspirin be synthesized from benzene?b. Ibuprofen is the active ingredient in pain relievers such as Advil, Motrin and Nuprin. How can ibuprofen be synthesized from benzene?c. Acetaminophen is the active ingredient in Tylenol. How can acetaminophen be synthesized from benzene?
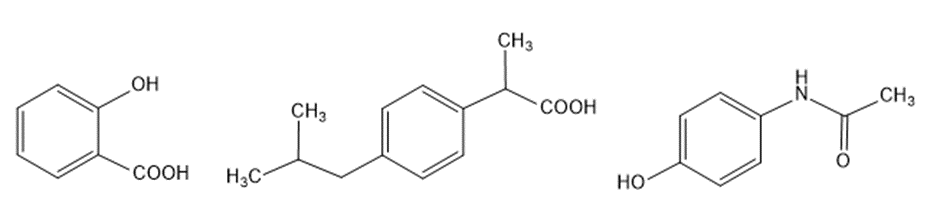
aspirin ibuprofen acetaminophen
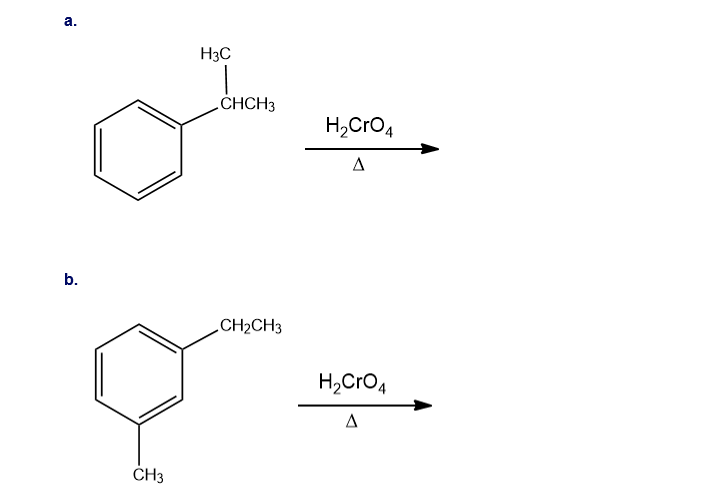
What are the major products of the following reactions?

a. Rank the following compounds from greatest tendency to least tendency to undergo nucleophilic aromatic substitution:
chlorobenzene 1-chloro-2,4-dinitrobenzene p-chloronitrobenzene
b. Rank the same compounds from greatest tendency to least tendency to undergo electrophilic aromatic substitution.
Draw resonance contributors for the carbanion that would be formed if meta-chloronitrobenzene were to react with hydroxide ion. Why doesn’t the reaction occur?
Draw resonance contributors for the carbanion that would be formed if meta-chloronitrobenzene were to react with hydroxide ion. Why doesn’t the reaction occur?
What do you think about this solution?
We value your feedback to improve our textbook solutions.