Chapter 18: Q 105 P (page 923)
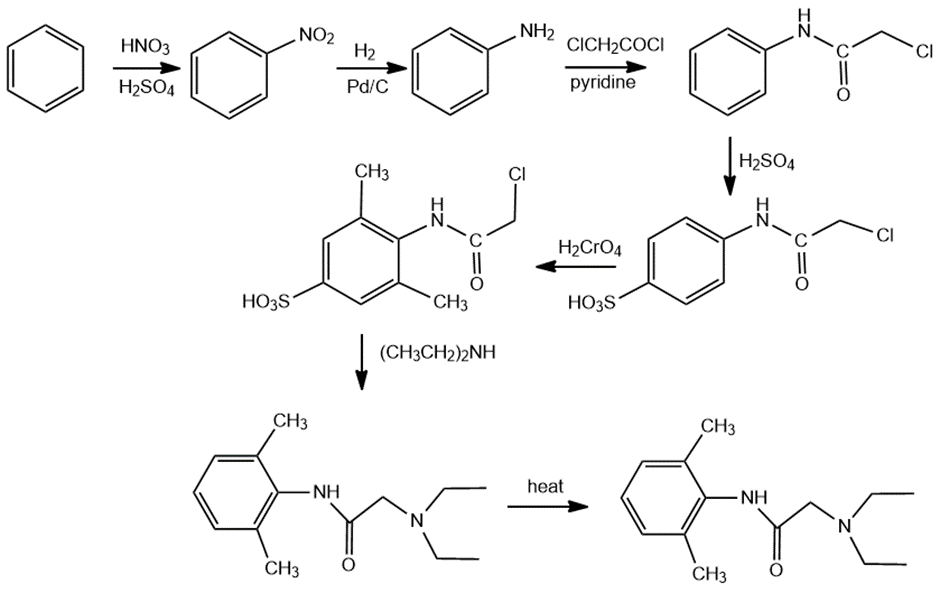
Question: Show how lidocain, one of the most widely used injectable anesthetics, can be prepared from benzene and compounds containing no more than four carbons.
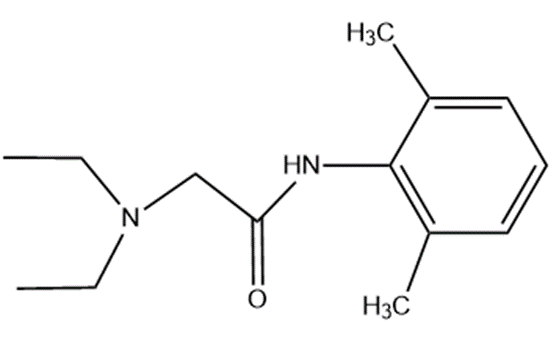
lidocain
Short Answer
The answer is,
Learning Materials
Features
Discover
Chapter 18: Q 105 P (page 923)
Question: Show how lidocain, one of the most widely used injectable anesthetics, can be prepared from benzene and compounds containing no more than four carbons.

lidocain
The answer is,

All the tools & learning materials you need for study success - in one app.
Get started for free
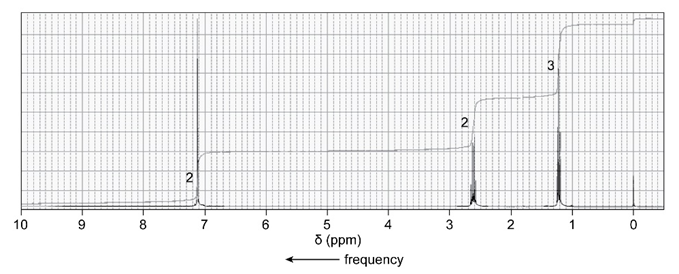
An unknown compound reacts with ethyl chloride and aluminum trichloride to form a compound that has the following 1H NMR spectrum. What is thestructure of the compound?
A mixture of 0.10 mol benzene and 0.10 mol p-xylene was allowed to react with 0.10 mol nitronium ion until all the nitronium ion was gone. Two products were obtained: 0.002 mol of one and 0.098 mol of the other.
What product is formed from the reaction of p-methylphenol with benzenediazonium chloride?
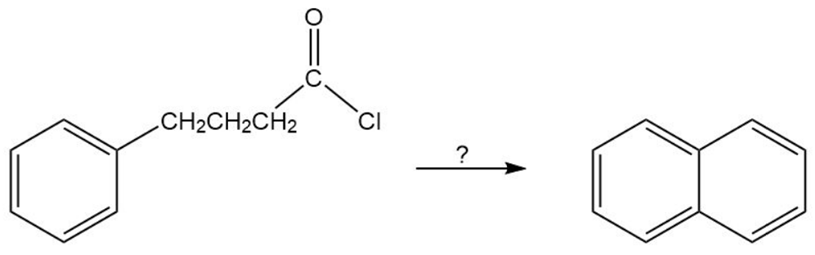
Describe how naphthalene can be prepared from the given starting material.
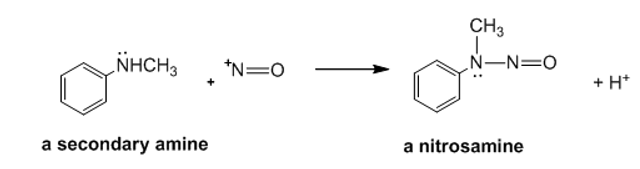
Explain why a secondary amine forms a nitrosamine rather than a diazonium ion when it reacts with a nitrosonium ion.
What do you think about this solution?
We value your feedback to improve our textbook solutions.