Chapter 16: Q 85P (page 797)
Propose a mechanism for each of the following reactions:
Short Answer
Expert verified
Answer:
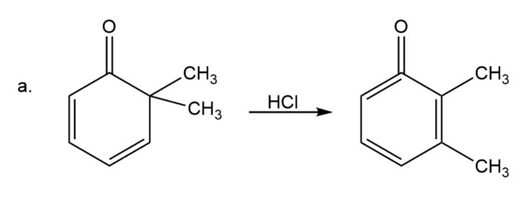
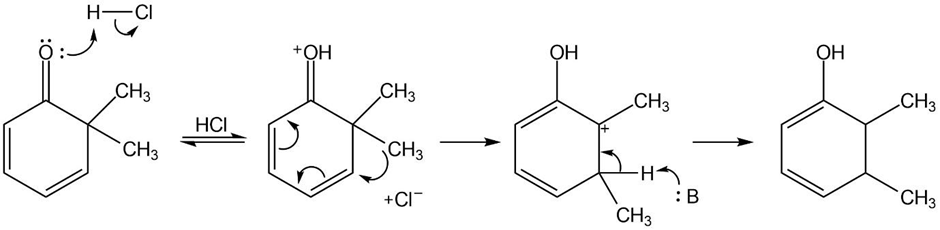
(a)
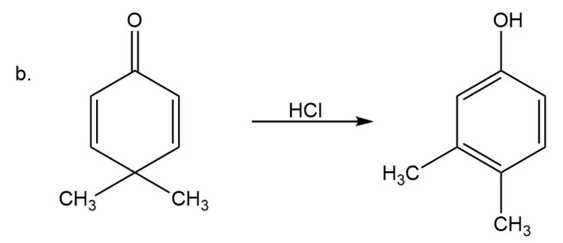
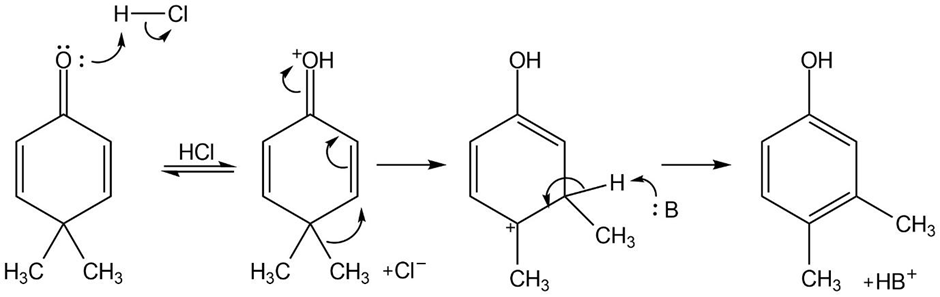
(b)
Learning Materials
Features
Discover
Chapter 16: Q 85P (page 797)
Propose a mechanism for each of the following reactions:


Answer:
(a)

(b)

All the tools & learning materials you need for study success - in one app.
Get started for free
What are the products of the following reactions?
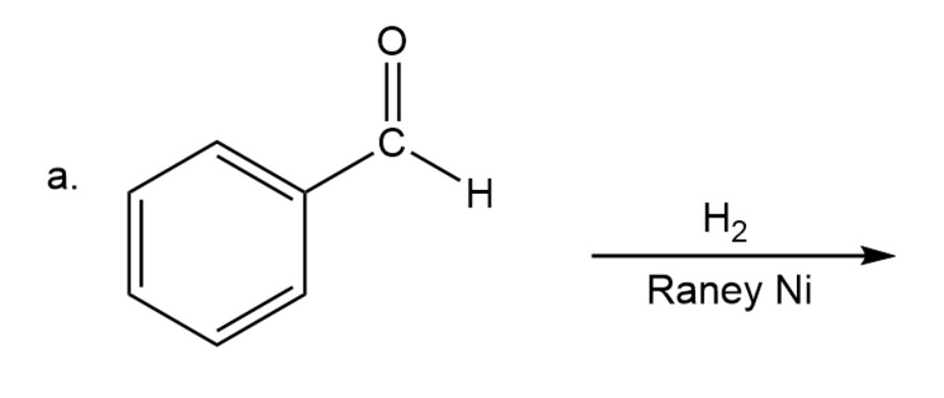
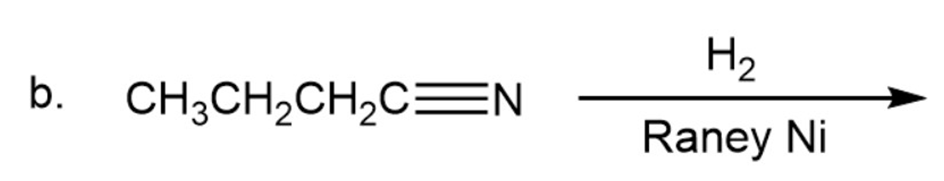
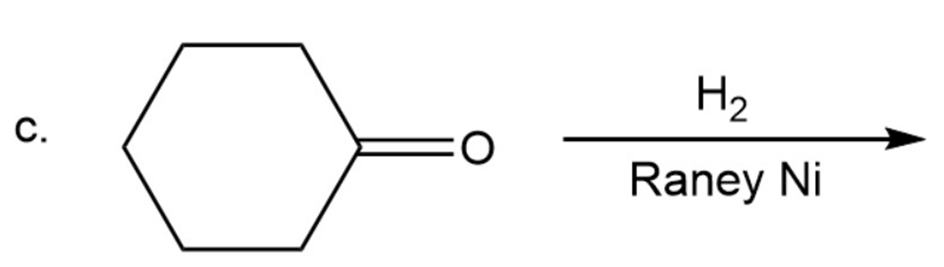
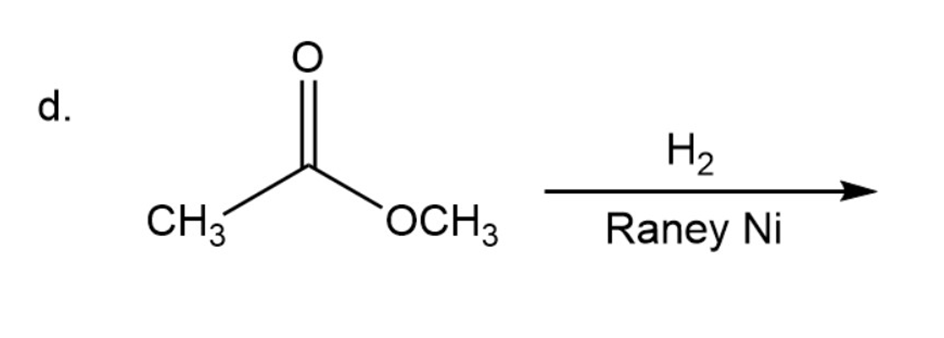
What are the products of the following reactions? Show all stereoisomers that are formed.
a)
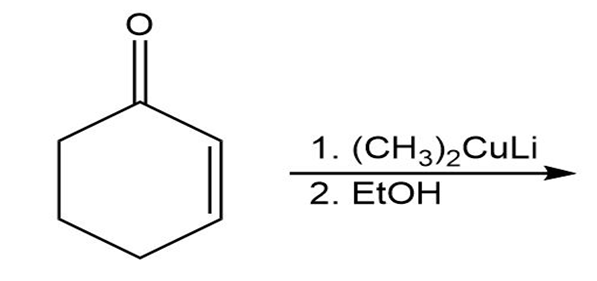
b)
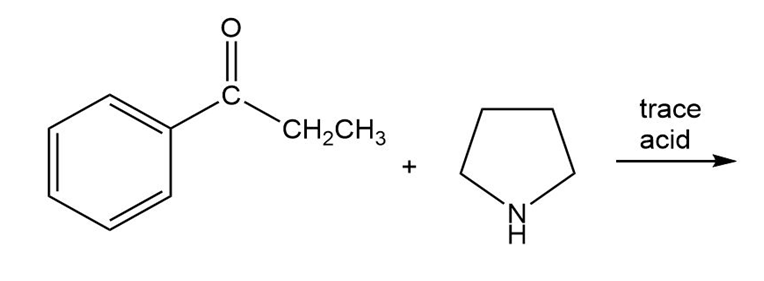
c)
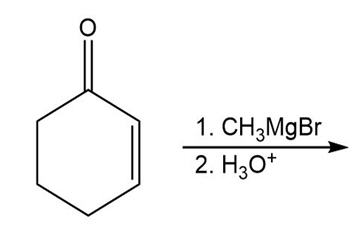
d)
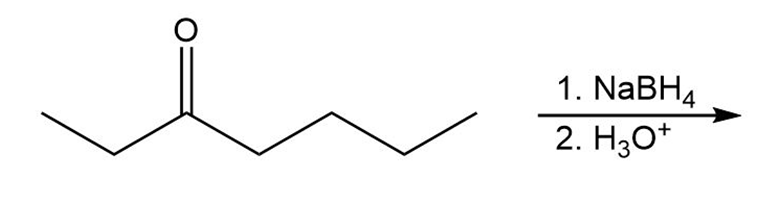
What is the product of the reaction of an ester with excess acetylide ion followed by the addition of pyridinium chloride?
Explain why an acetal can be isolated but most hydrates cannot be isolated.
Question: Name the following:
a.
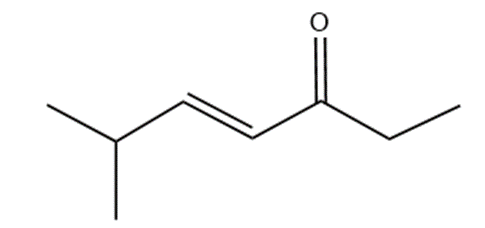
b.
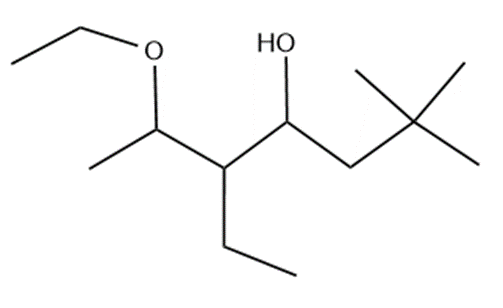
c.
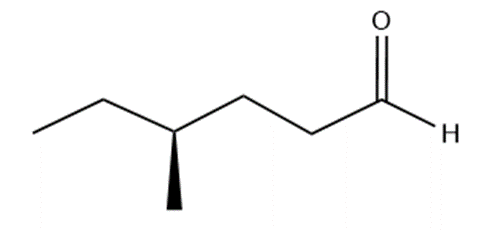
d.
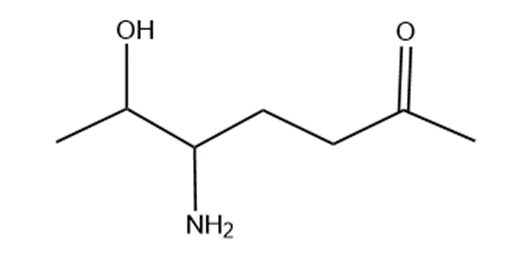
What do you think about this solution?
We value your feedback to improve our textbook solutions.