Chapter 10: Q5P (page 463)
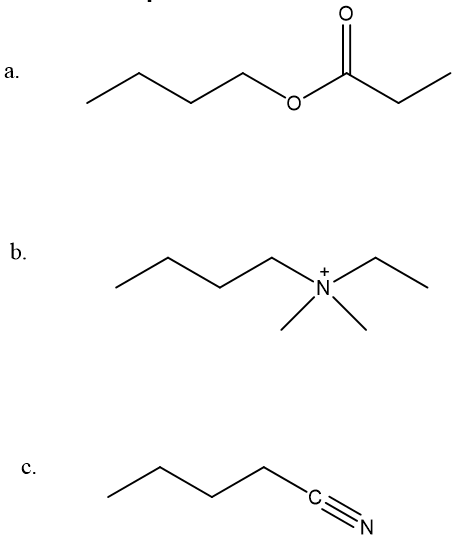
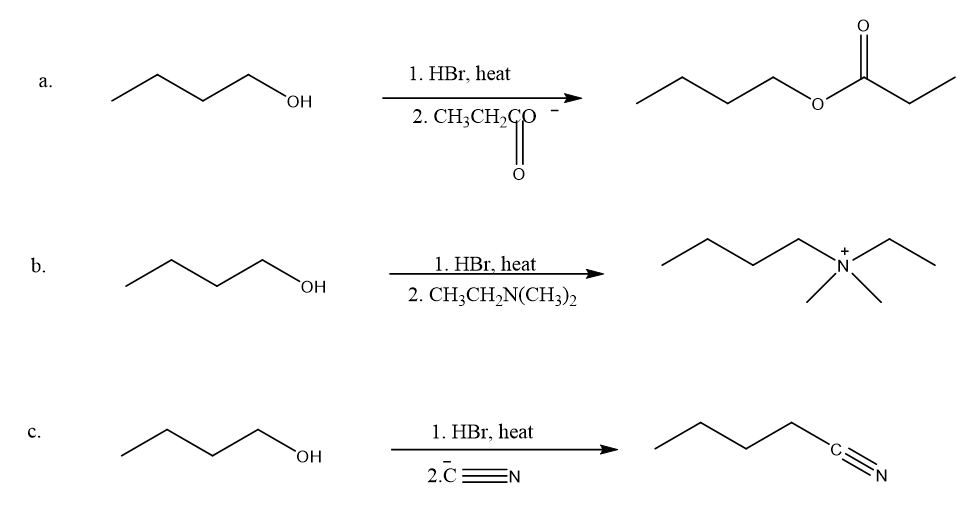
Explain how 1-butanol can be converted into the following compounds:
Short Answer
Learning Materials
Features
Discover
Chapter 10: Q5P (page 463)
Explain how 1-butanol can be converted into the following compounds:


All the tools & learning materials you need for study success - in one app.
Get started for free
Heating an alcohol with sulfuric acid is a good way to prepare a symmetrical ether such as diethyl ether.
a. Explain why it is not a good way to prepare an unsymmetrical ether such as ethyl propyl ether.
b. How would you synthesize ethyl propyl ether?
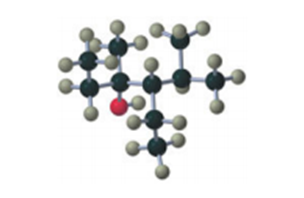
If the compound shown in the margin is heated in the presence of H2SO4,
a. what constitutional isomer would be formed in greatest yield?
b. what stereoisomer would be formed in greater yield?
What product is obtained from the reaction of each of the following alcohols with
a. H2CrO4? b. HOCl? c. the regents required for a Swern oxidation?
1. 3-pentanol
2. 1-pentanol
3. 2-methyl-2-pentanol
4. 2,4-hexanediol
5. cyclohexanol
6. 1,4-butanediol
What stereoisomers are formed in the following reactions? Which stereoisomer is the major product?
a. the acid-catalyzed dehydration of 1-pentanol to 2-pentene
b. the acid-catalyzed dehydration of 3,4-dimethyl-3-hexanol to 3,4-dimethyl-3-hexene
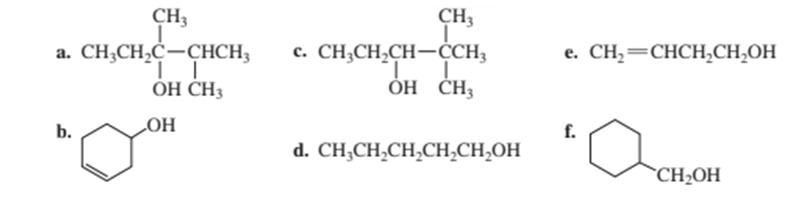
What is the major product obtained when each of the following alcohols is heated in the presence of H2SO4?
What do you think about this solution?
We value your feedback to improve our textbook solutions.