Chapter 10: Q12P (page 468)
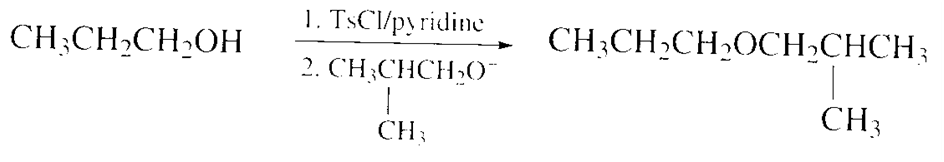
Show how 1-propanol can be converted into the following compounds by means of a sulfonate ester:

Short Answer
a)

b)
Learning Materials
Features
Discover
Chapter 10: Q12P (page 468)
Show how 1-propanol can be converted into the following compounds by means of a sulfonate ester:

a)

b)

All the tools & learning materials you need for study success - in one app.
Get started for free
Explain the difference in reactivity between CH3OH2 and CH3OH in a nucleophilic substitution reaction. (The pKa of H3O+ is -1.7.)
If the compound shown in the margin is heated in the presence of H2SO4,
a. what constitutional isomer would be formed in greatest yield?
b. what stereoisomer would be formed in greater yield?

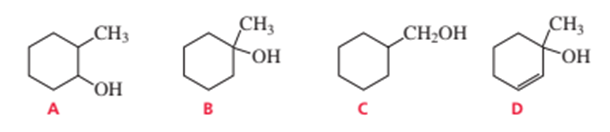
Which of the following alcohols dehydrates the fastest when heated with acid?
What stereoisomers do the following reactions form?

What stereoisomers are formed in the following reactions? Which stereoisomer is the major product?
a. the acid-catalyzed dehydration of 1-pentanol to 2-pentene
b. the acid-catalyzed dehydration of 3,4-dimethyl-3-hexanol to 3,4-dimethyl-3-hexene
What do you think about this solution?
We value your feedback to improve our textbook solutions.