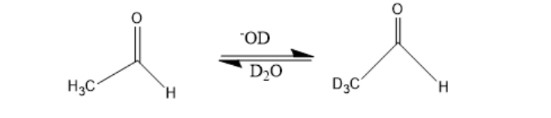
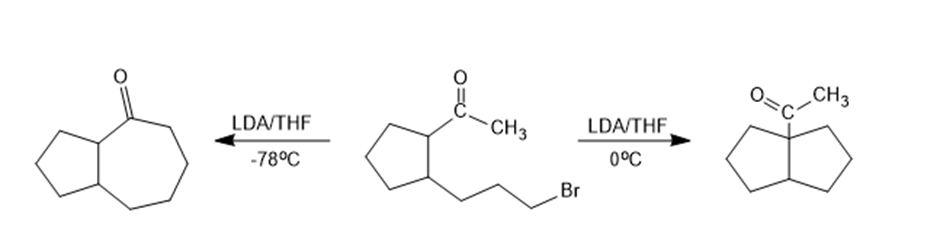
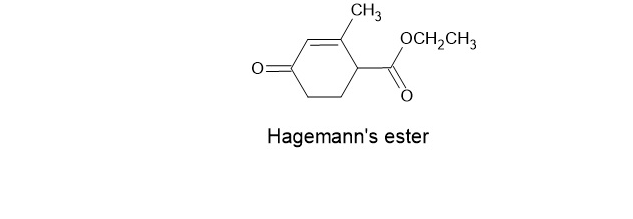
Chapter 17: Q3P (page 804)
Explain why a base can remove a proton from the -carbon of N,N-dimethylethanamide but not from the -carbon of either N-methylethanamide or ethanamide.
N,N-dimethylethanamideN-methylethanamide Ethanamide
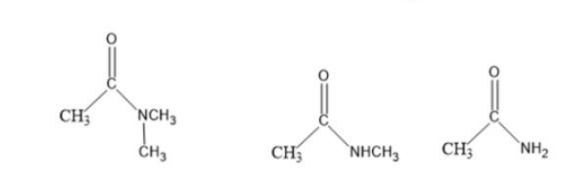
Short Answer
Due to the lack of N-H bond in N, N-dimethyl ethanamide, a base can remove a proton from the alpha carbon of N, N-dimethyl ethanamine.
 Removal of proton
Removal of proton Removal of proton
Removal of proton