Chapter 19: Q8P (page 933)
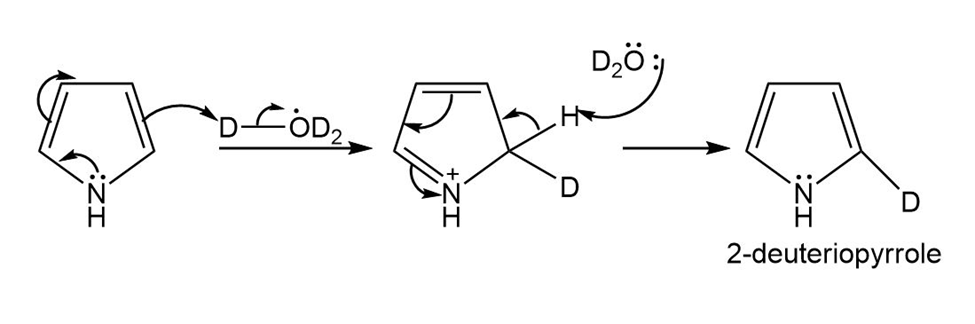
When pyrrole is added to a dilute solution of in , 2-deuteriopyrrole is formed. Propose a mechanism to account for the formation of this compound.
Short Answer
Learning Materials
Features
Discover
Chapter 19: Q8P (page 933)
When pyrrole is added to a dilute solution of in , 2-deuteriopyrrole is formed. Propose a mechanism to account for the formation of this compound.

All the tools & learning materials you need for study success - in one app.
Get started for free
Why is imidazole a stronger acid (pKa = 14.4) than pyrrole (pKa~ 17)?
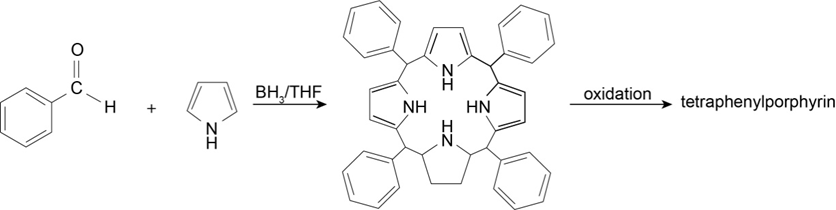
Organic chemists work with tetraphenylporphyrins rather than with porphyrins because tetraphenylporphyrins are much more resistant to air oxidation. Tetraphenylporphyrin can be prepared by the reaction of benzaldehyde with pyrrole. Propose a mechanism for the formation of the ring system shown here:
a.Draw the structure of 3-quinuclidinone.
b.What is the approximateof its conjugate acid?
c.Which has a lowervalue, the conjugate acid of 3-bromoquinuclidine or the conjugate acid of 3-chloroquinuclidine?
The dipole moments of furan and tetrahydrofurZan are in the same direction. One compound has a dipole moment of 0.70 D, and the other has a dipole moment of 1.73 D. Which is which?
Why is protonated pyrimidine more acidic than protonated pyridine ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.