Chapter 19: Q6P (page 930)
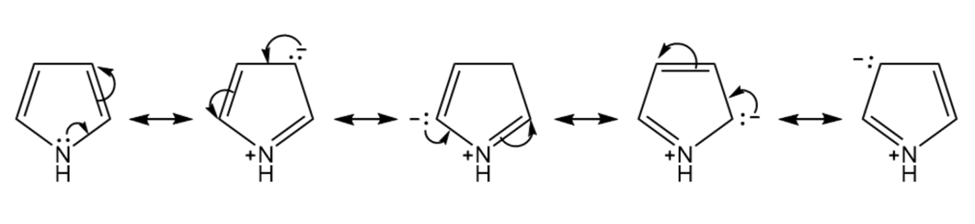
Draw arrows to show the movement of electrons in going from one resonance contributor of pyrrole to the next.
Short Answer
Learning Materials
Features
Discover
Chapter 19: Q6P (page 930)
Draw arrows to show the movement of electrons in going from one resonance contributor of pyrrole to the next.

All the tools & learning materials you need for study success - in one app.
Get started for free
Rank imidazole, pyrrole, and benzene from most reactive to least reactive toward electrophilic aromatic substitution.
One of the following compounds undergoes electrophilic aromatic substitution predominantly at C-3, and one undergoes electrophilic aromatic substitution predominantly at C-4. Which is which?
The chemical shifts of the C-2 hydrogen in the spectra of pyrrole, pyridine, and pyrrolidine are 2.82 ppm, 6.42 ppm, and 8.50 ppm. Match each heterocycle with its chemical shift.
What are the products of the following reactions?
a.
b.
c.
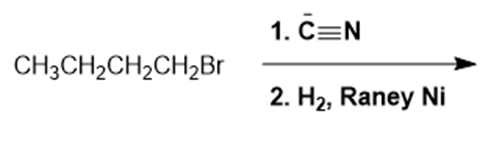
d.
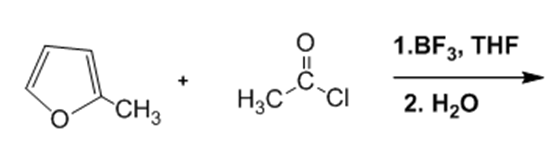
e.
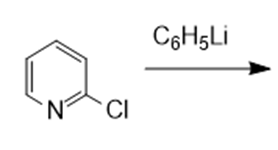
f.
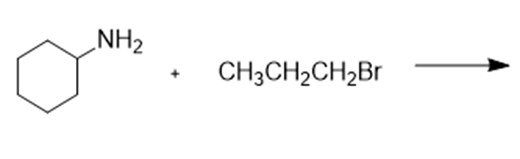
g.
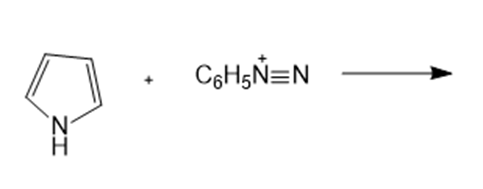
h.
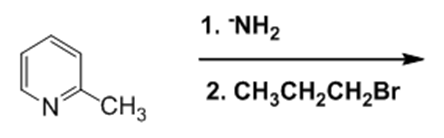
i.
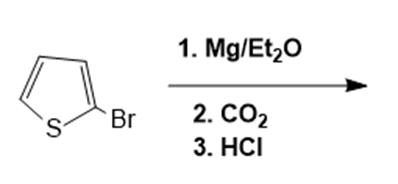
Draw the product of each of the following reactions:
a.
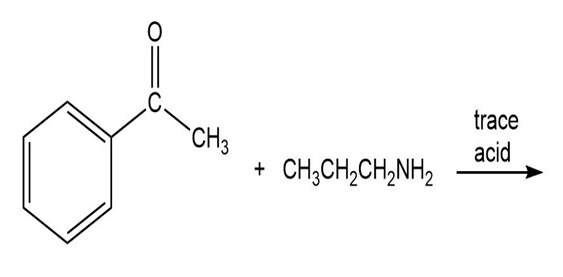
b.
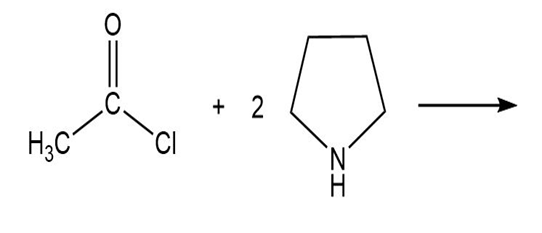
C.
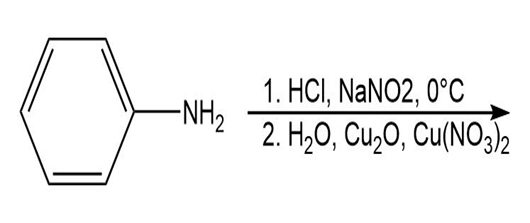
d.
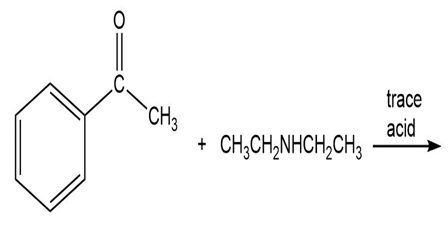
What do you think about this solution?
We value your feedback to improve our textbook solutions.