Chapter 19: Q18P (page 941)
What percent of imidazole is protonated at physiological pH (7.4)?
Short Answer
The percentage of imidazole protonated at physiological pH (7.4) is 20 %.
Learning Materials
Features
Discover
Chapter 19: Q18P (page 941)
What percent of imidazole is protonated at physiological pH (7.4)?
The percentage of imidazole protonated at physiological pH (7.4) is 20 %.
All the tools & learning materials you need for study success - in one app.
Get started for free
Name the following:
a.
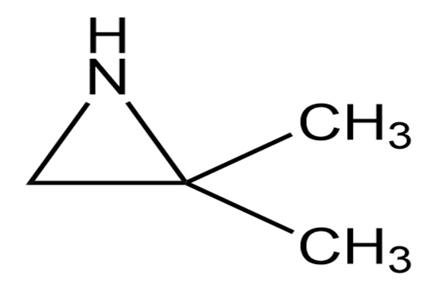
b.
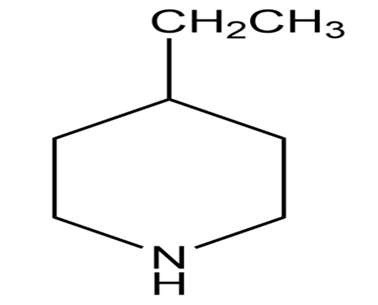
c.
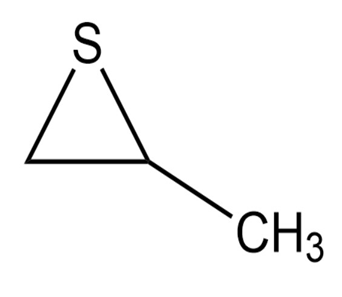
d.
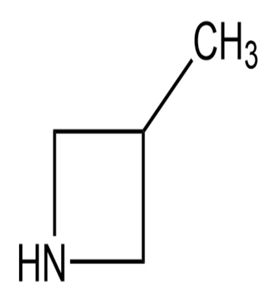
e.
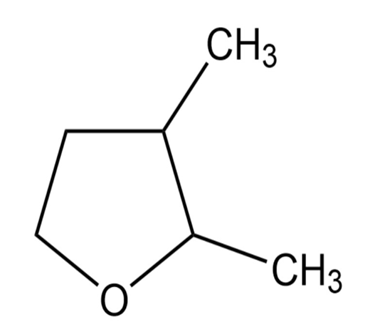
f.
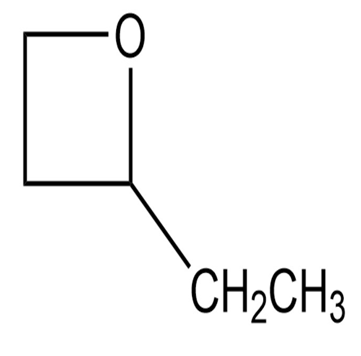
Why is imidazole a stronger acid (pKa = 14.4) than pyrrole (pKa~ 17)?
Draw the product of each of the following reactions:
a.
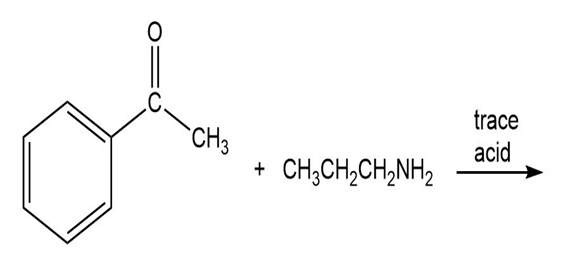
b.
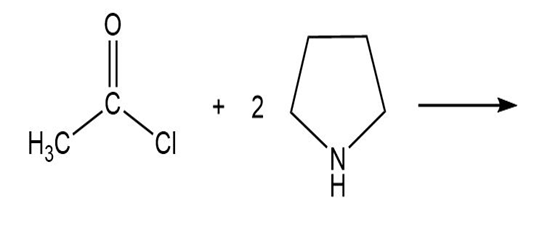
C.
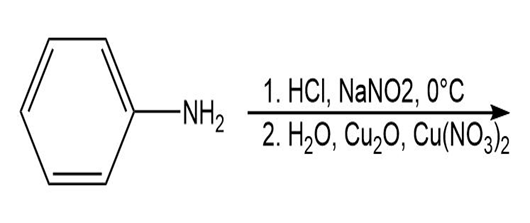
d.
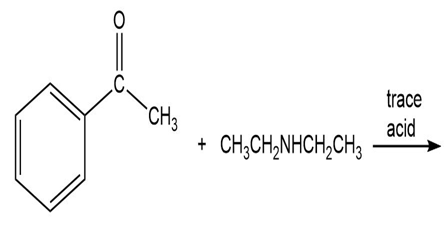
a. Draw resonance contributors to show why pyridine-N-oxide is more reactive than pyridine toward electrophilic aromatic substitution.
b. At what position does pyridine-N-oxide undergo electrophilic aromatic substitution?
Question: Name the following:
a.
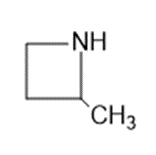
b.
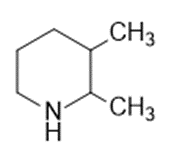
c.
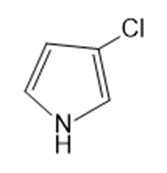
d.
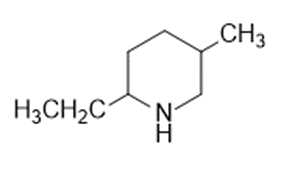
What do you think about this solution?
We value your feedback to improve our textbook solutions.