Chapter 4: Q41P (page 166)
Draw the stereoisomers of the following amino acids. Indicate pairs of enantiomers and pairs of diastereomers.

Short Answer
a. 2 stereoisomers- S and R
b. 4 stereoisomers- RR, SS, RS and SR.
Learning Materials
Features
Discover
Chapter 4: Q41P (page 166)
Draw the stereoisomers of the following amino acids. Indicate pairs of enantiomers and pairs of diastereomers.

a. 2 stereoisomers- S and R
b. 4 stereoisomers- RR, SS, RS and SR.
All the tools & learning materials you need for study success - in one app.
Get started for free
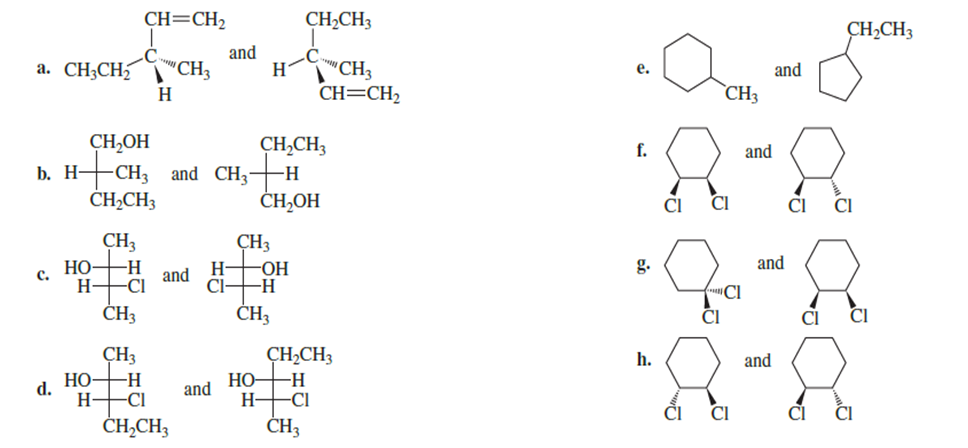
Question:Are the following pairs identical, enantiomers, diastereomers, or constitutional isomers?
1-Bromo-2-methylcyclopentane has four pairs of diastereomers. Draw the four pairs
Naproxen, a nonsteroidal anti-inflammatory drug that is the active ingredient in Aleve (p. 115), has a specific rotation of +66. One commercial preparation result in a mixture with a 97% enantiomeric excess.
a. Does naproxen have the R or the S configuration?
b. What percent of each enantiomer is obtained from the commercial preparation?
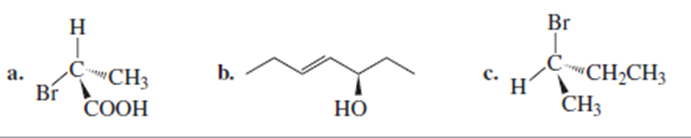
Do the following compounds have the R or the S configurations?
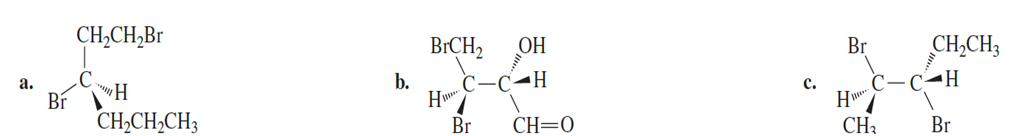
Question:What is the configuration of the asymmetric centers in the following compounds?
What do you think about this solution?
We value your feedback to improve our textbook solutions.