Chapter 4: Q16P (page 153)
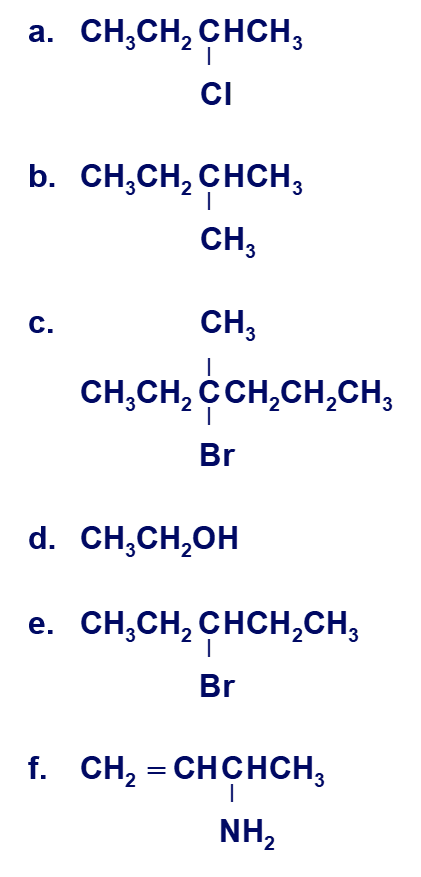
Which of the compounds in Problem 14 can exist as enantiomers?
Short Answer
The compounds that can exist as an enantiomer are:
a)Enantiomer
b) Not Enantiomer
c)Enantiomer
d) Not Enantiomer
e) Not Enantiomer
f)Enantiomer
Learning Materials
Features
Discover
Chapter 4: Q16P (page 153)
Which of the compounds in Problem 14 can exist as enantiomers?

The compounds that can exist as an enantiomer are:
a)Enantiomer
b) Not Enantiomer
c)Enantiomer
d) Not Enantiomer
e) Not Enantiomer
f)Enantiomer
All the tools & learning materials you need for study success - in one app.
Get started for free
Naproxen, a nonsteroidal anti-inflammatory drug that is the active ingredient in Aleve (p. 115), has a specific rotation of +66. One commercial preparation result in a mixture with a 97% enantiomeric excess.
a. Does naproxen have the R or the S configuration?
b. What percent of each enantiomer is obtained from the commercial preparation?
a.Which of the following compounds can exist as cis-trans isomers?
b. For those compounds that can exist as cis and trans isomers, draw and label the isomers.
1 CH3CH = CHCH2CH2CH3
2 CH3CH2 C = CHCH3
|
CH2CH3
3.CH3CH = CHCH3
4. CH3CH2CH=CH2
Draw the stereoisomers of the following amino acids. Indicate pairs of enantiomers and pairs of diastereomers.
What is the configuration of the following compounds? (Use the given structures to answer the question.)
a. (-)-glyceric acid
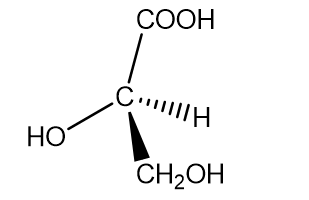
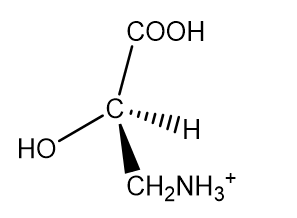
b.(+)-isoserine
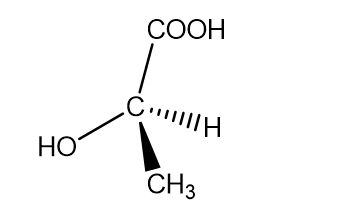
c. (+)-lactic acid
What do you think about this solution?
We value your feedback to improve our textbook solutions.