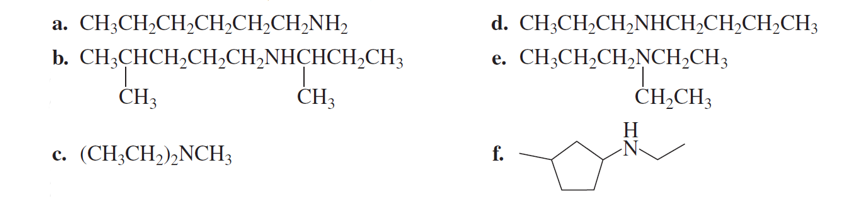Chapter 3: Q70P (page 138)
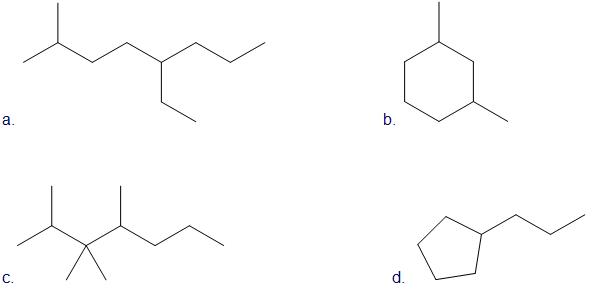
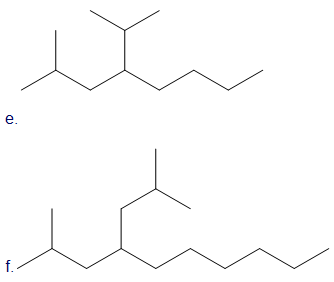
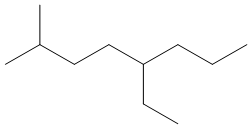
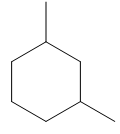
Draw skeletal structures for the following:
a. 5-ethyl-2-methyloctane
b. 1,3-dimethylcyclohexane
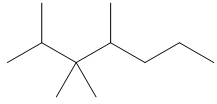
c. 2,3,3,4-tetramethylheptane
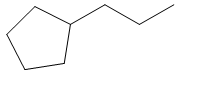
d. propyl cyclopentane
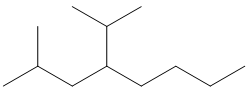
e. 2-methyl-4-(1-methylethyl) octane
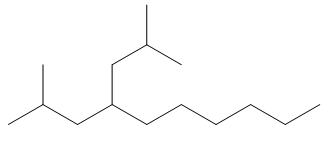
f. 2,6-dimethyl-4-(2-methylpropyl)decane
Short Answer