Chapter 3: Q68P (page 138)
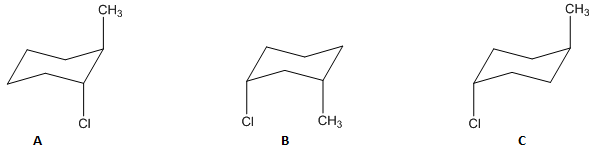
Which of the following conformers has the highest energy (is the least stable)?
Short Answer
Compound B has the highest energy and is the least stable.
Learning Materials
Features
Discover
Chapter 3: Q68P (page 138)
Which of the following conformers has the highest energy (is the least stable)?

Compound B has the highest energy and is the least stable.
All the tools & learning materials you need for study success - in one app.
Get started for free
a. Calculate the energy difference between the two chair conformers of trans-1,4-dimethylcyclohexane.
b. What is the energy difference between the two chair conformers of cis-1,4-dimethylcyclohexane?
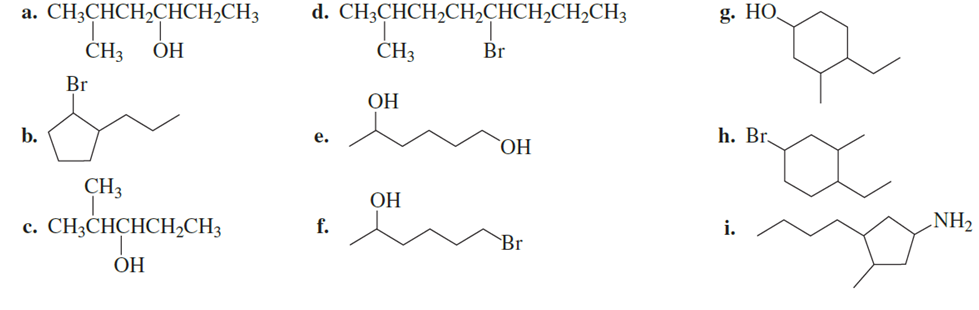
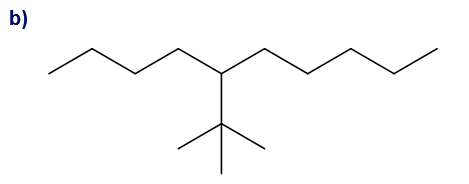
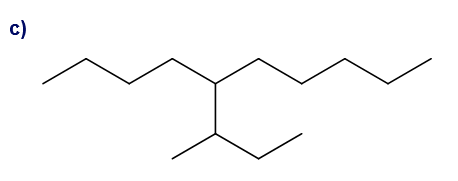
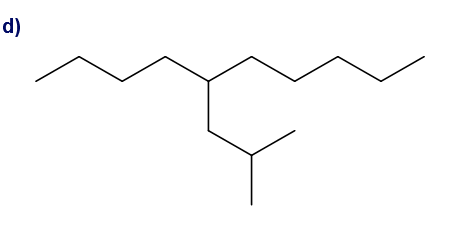
What is each compound’s systematic name?
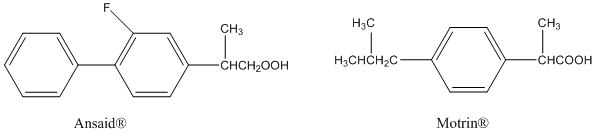
Ansaid and Motrin belong to the group of drugs known as nonsteroidal anti-inflammatory drugs (NSAIDs). Both are only slightly soluble in water, but one is a little more soluble than the other. Which of the drugs has the greater solubility in water?
One of the chair conformers of cis-1,3-dimethylcyclohexane is 5.4 kcal/mol less stable than the other. How much steric strain does a 1,3-diaxial interaction between two methyl groups introduce into the conformer?
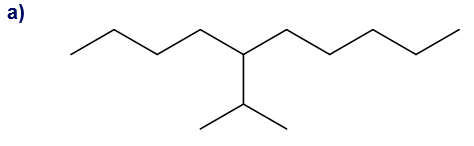
Give each substituent on the nine-carbon chain a common name and a parenthetical name.
What do you think about this solution?
We value your feedback to improve our textbook solutions.