Chapter 3: Q56 P (page 136)
List the following compounds from highest boiling to lowest boiling:
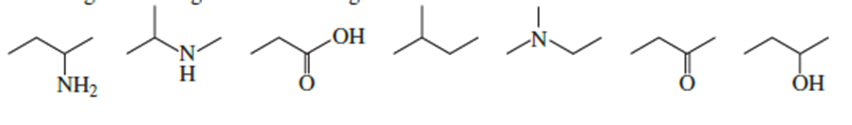
Short Answer
Arrangement of compounds from the highest boiling point to the lowest.
Learning Materials
Features
Discover
Chapter 3: Q56 P (page 136)
List the following compounds from highest boiling to lowest boiling:

Arrangement of compounds from the highest boiling point to the lowest.

All the tools & learning materials you need for study success - in one app.
Get started for free
Write condensed and skeletal structures for all the tertiary alcohols with molecular formula C6H14O and give each a systematic name.
What is each compound’s systematic name?
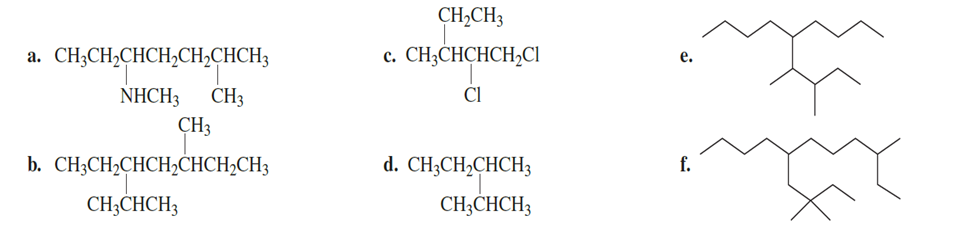
Draw the structure for each of the following:
a. 2,2-dimethyl-4-isopropyloctane
b. 2,3-dimethylhexane
c. 4,4-diethyldecane
d. 2,4,5-trimethyl-4-(1-methylethyl) heptane
e. 2,5-dimethyl-4-(2-methylpropyl) octane
f. 4-(1,1-dimethylethyl) octane
Draw the structures of a homologous series of alcohols that have from one to six carbons and give each of them a common name and a systematic name.
Draw the structures and provide systematic names for parts a, b, and c by substituting a chlorine for a hydrogen of methylcyclohexane:
a. A primary alkyl halide
b. A tertiary alkyl halide
c. Three secondary alkyl halides
What do you think about this solution?
We value your feedback to improve our textbook solutions.