Chapter 3: Q.3-35P (page 114)
Rank the following compounds from highest boiling to lowest boiling:
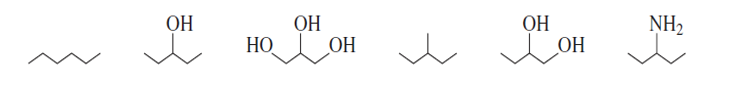
Short Answer
Ranking from highest boiling to lowest boiling is as follows,
3>5>2>6>1>4.

Due to the H bonding and branching in the main straight chain.
Learning Materials
Features
Discover
Chapter 3: Q.3-35P (page 114)
Rank the following compounds from highest boiling to lowest boiling:
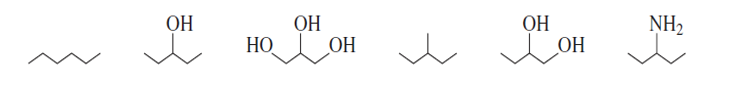
Ranking from highest boiling to lowest boiling is as follows,
3>5>2>6>1>4.

Due to the H bonding and branching in the main straight chain.
All the tools & learning materials you need for study success - in one app.
Get started for free
A student was given the structural formulas of several compounds and was asked to give them systematic names. How many did the student name correctly? Correct those that are misnamed.
l. 2-methyl-N, N-dimethyl-4-hexanamine
Draw a condensed structure and a skeletal structure for each of the following:
a. sec-butyl tert-butyl ether
b. isoheptyl alcohol
c. sec-butylamine
d. isopentyl bromide
e. 5-(1-methylethyl)nonane
f. triethylamine
g. 4-(1,1-dimethylethyl)heptane
h. 5,5-dibromo-2-methyloctane
i. 3-ethoxy-2-methylhexane
j. 5-(1,2-dimethylpropyl)nonane
k. 3,4-dimethyloctane
l. 5-isopentyldecane
Write condensed and skeletal structures for all the tertiary alcohols with molecular formula C6H14O and give each a systematic name.
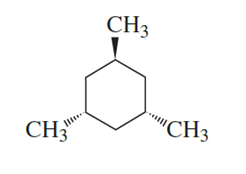
Draw the most stable conformer of the following molecule. (A solid wedge points out of the plane of the paper toward the viewer. A hatched wedge points back from the plane of the paper away from the viewer.)
Predict the approximate size of the following bond angles.
a. the C-O-C bond angle in an ether
b. the C-N-C bond angle in a secondary amine
c. the C-O-H bond angle in an alcohol
d. the C-N-C bond angle in a quaternary ammonium salt
What do you think about this solution?
We value your feedback to improve our textbook solutions.