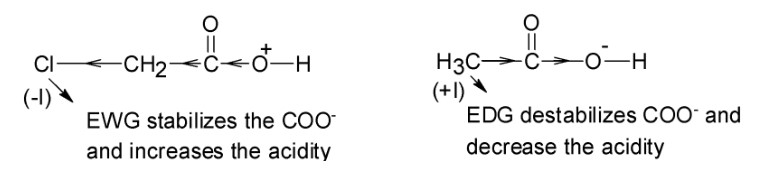Chapter 21: Q9P (page 995)
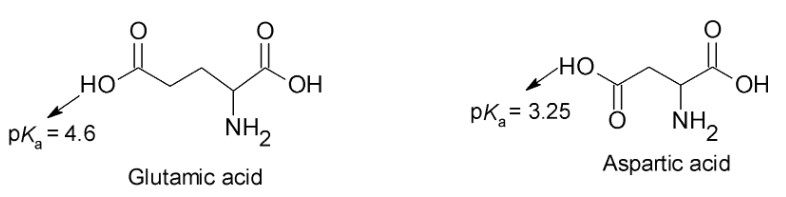
a. Why is the pKaof the glutamate side chain greater than the pKaof the aspartate side chain?
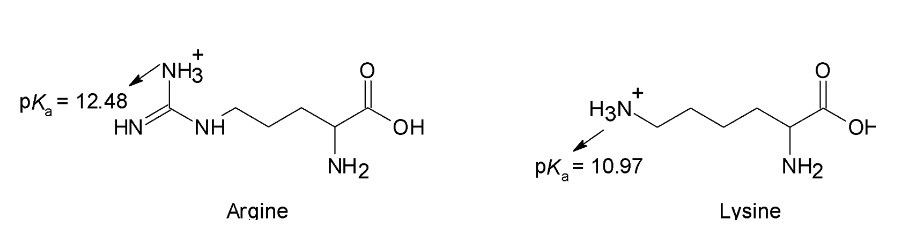
b. Why is the pKa of the arginine side chain greater than the pKa of the lysine side chain?
Short Answer
a. The pKaof the glutamate side chain greater than the pKaof the aspartate side chain because the carboxyl group of the aspartic acid side chain is stronger acid than carboxyl group of the glutamic acid side chain because carboxyl group of aspartic acid side chain is closer to electron-withdrawing protonated amino group.
b. The pKa of the arginine side chain greater than the pKa of the lysine side chain because the protonated lysine side chain is stronger acid than the protonated arginine side chain due to less of tendency of arginine side chain to lose a proton as its positive charge is delocalized over three nitrogen atoms.