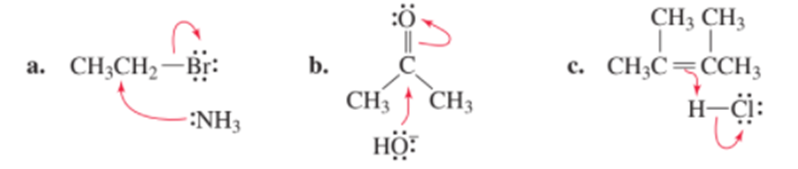
Chapter 5: Q59P (page 223)
a. Which of the following reactions will have the larger ΔS° value?
b. Will the ΔS° value be positive or negative?\
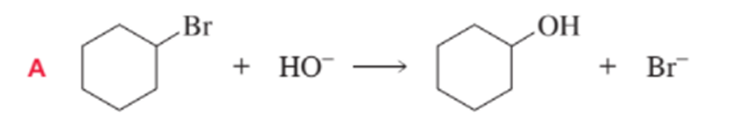
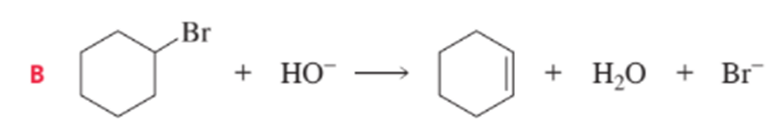
Short Answer
a) B will have the largerΔS°value. As in A reactant and product are same but in B these are different.
b) In B Product has greater freedom of motion then it has ΔS° value positive.