Chapter 2: Q59P (page 78)
Rank the following compounds from strongest to weakest acid
CH3CH2OH CH3CH2NH2 CH3CH2SH CH3CH2CH3
Short Answer
The decreasing order of strength of acids is:
CH3CH2SH > CH3CH2OH> CH3CH2CH3> CH3CH2NH2
Learning Materials
Features
Discover
Chapter 2: Q59P (page 78)
Rank the following compounds from strongest to weakest acid
CH3CH2OH CH3CH2NH2 CH3CH2SH CH3CH2CH3
The decreasing order of strength of acids is:
CH3CH2SH > CH3CH2OH> CH3CH2CH3> CH3CH2NH2
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:Draw the products of the acid–base reaction when
a. HCl is the acid and NH3 is the base.
b. H2O is the acid and -NH2 is the base.
You are planning to carry out a reaction that produces protons. The reaction will be buffered at pH = 10.5. Would it be better to use a protonated methylamine/methylamine buffer or a protonated ethylamine/ethylamine buffer? (pKa of protonated methylamine = 10.7; pKaof protonated ethylamine = 11.0)
Butyric acid, the compound responsible for the unpleasant odor and taste of sour milk, has a pKa value of 4.82. What is its Ka value? Is it a stronger acid or a weaker acid than vitamin C?
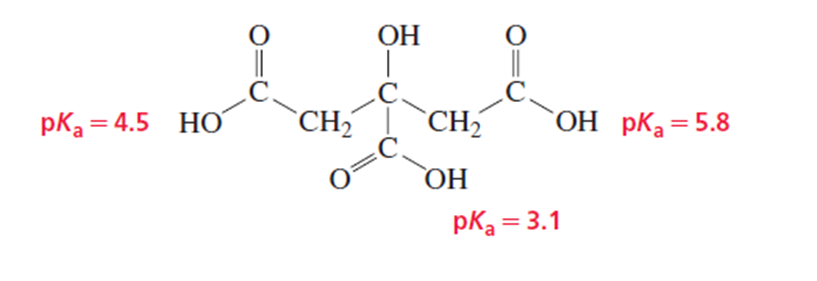
Citrus fruits are rich in citric acid, a compound with three COOH groups. Explain the following:
a. The first pKa (for the COOH group in the center of the molecule) is lower than the pKa of acetic acid.
b. The third pKa is greater than the pKa of acetic acid.
Explain why HCl has a smaller dipole moment than HF, even though the H-Cl bond is longer than the H-F bond.
What do you think about this solution?
We value your feedback to improve our textbook solutions.