Chapter 4: Q 45P (page 234)
Draw the important resonance forms of the following free radicals.
(a)
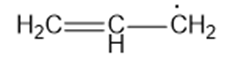
(b)
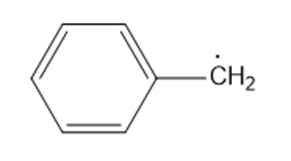
(c)
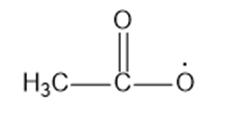
(d)
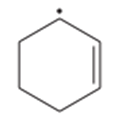
(e)
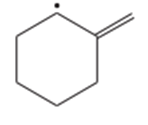
(f)
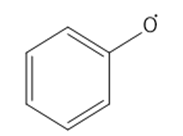
Short Answer
a)
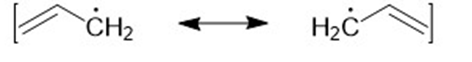
b)
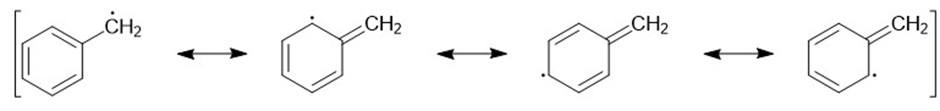
c)
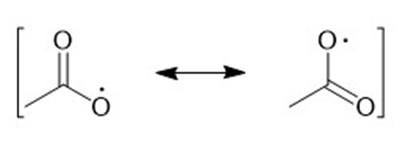
d)
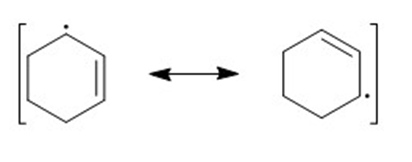
e)
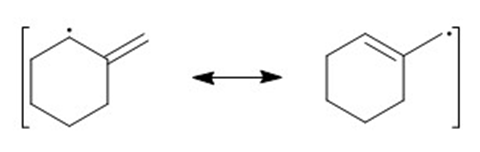
f)
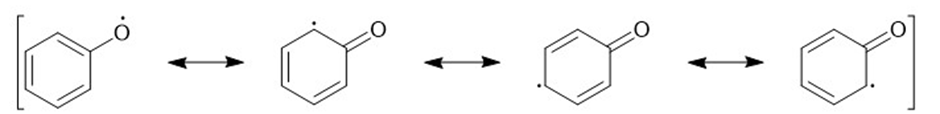
Learning Materials
Features
Discover
Chapter 4: Q 45P (page 234)
Draw the important resonance forms of the following free radicals.
(a)

(b)

(c)

(d)

(e)

(f)

a)

b)

c)

d)

e)

f)

All the tools & learning materials you need for study success - in one app.
Get started for free
When a small piece of platinum is added to a mixture of ethene and hydrogen, the following reaction occurs:
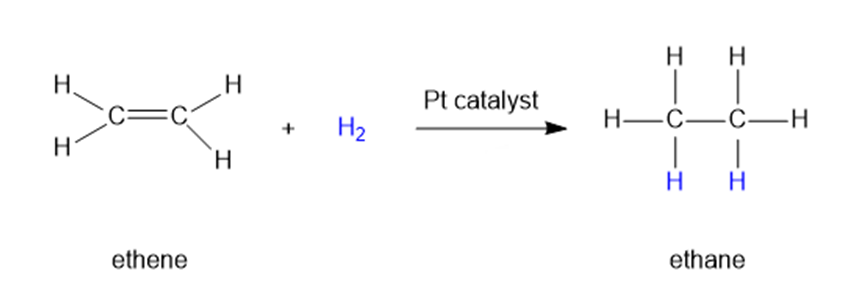
Doubling the concentration of hydrogen has no effect on the reaction rate. Doubling the concentration of ethene also has no effect.
(a) What is the kinetic order of this reaction with respect to ethene? With respect to hydrogen? What is the overall order?
(b) Write the unusual rate equation for this reaction.
(c)Explain this strange rate equation, and suggest what one might do to accelerate the reaction.
Use bond-dissociation enthalpies (Table 4-2, p.203) to calculate values ofΔHfor the following reactions.
(a)

(b)

(c)
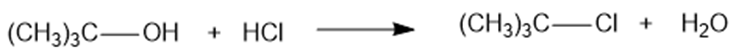
(d)

(e)

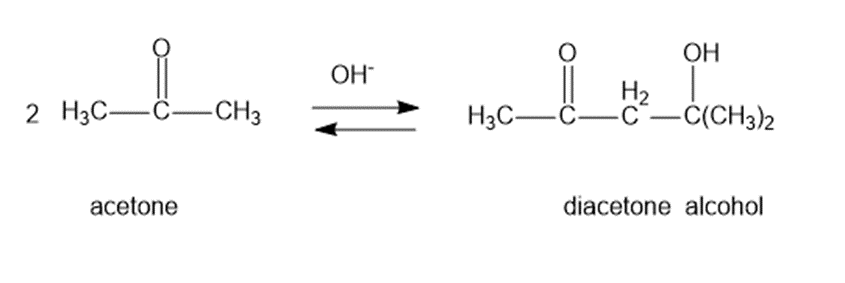
Question: Under base-catalyzed conditions, two molecules of acetone can condense to form diacetone alcohol. At room temperature, aboutof 5%the acetone is converted to diacetone alcohol. Determine the value offor this reaction.
Question: (a) Propose a mechanism for the free-radical chlorination of ethane.
(b) Calculatefor each step in this reaction
(c) Calculate the overall value offor this reaction.
Use the bond-dissociation enthalpies in Table 4-2 (page 203) to calculate the heats of reaction for the two possible first propagation steps in the chlorination of isobutane. Use this information to draw a reaction-energy diagram like Figure 4-8, comparing the activation energies of the two radicals.
What do you think about this solution?
We value your feedback to improve our textbook solutions.