Chapter 26: Q2P (page 1326)
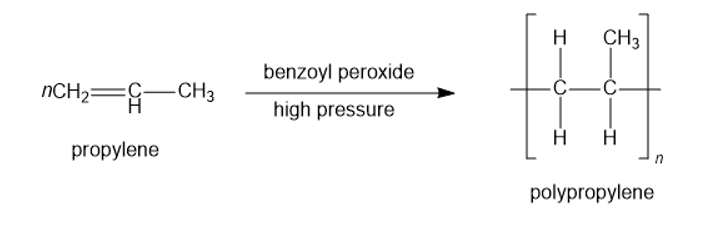
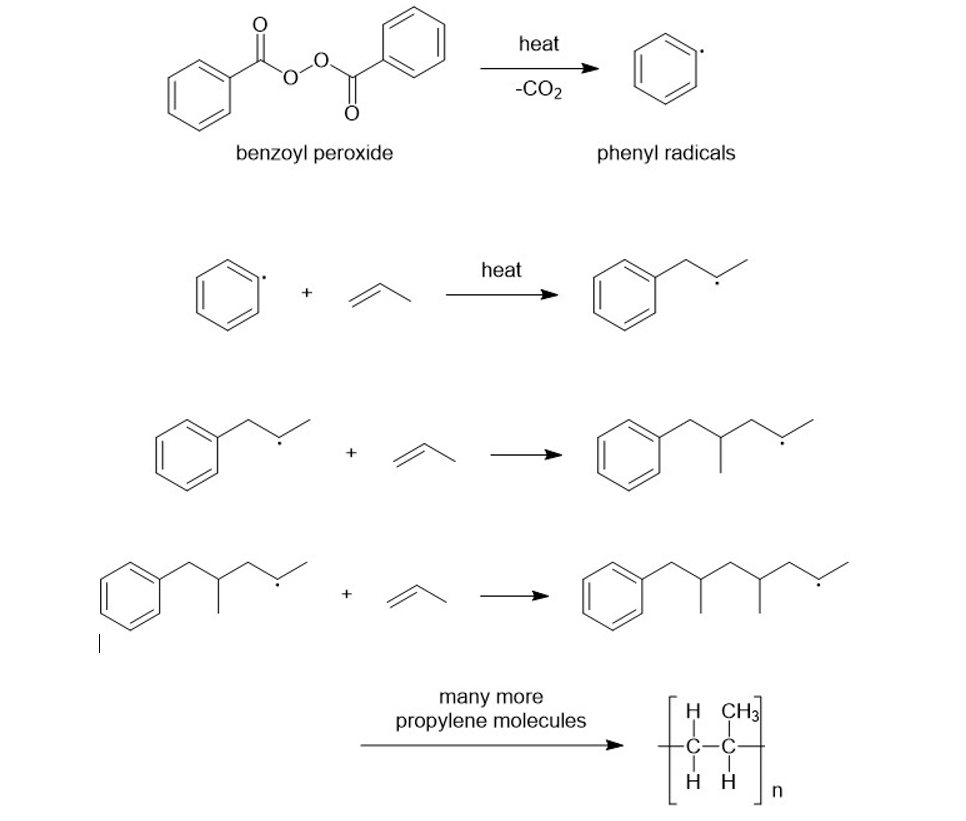
Propose a mechanism for reaction of the first three propylene units in the polymerization of propylene in the presence of benzoyl peroxide.
Short Answer
Learning Materials
Features
Discover
Chapter 26: Q2P (page 1326)
Propose a mechanism for reaction of the first three propylene units in the polymerization of propylene in the presence of benzoyl peroxide.


All the tools & learning materials you need for study success - in one app.
Get started for free
Bisphenol A is made on a large scale by a condensation of phenol with acetone. Suggest an appropriate catalyst, and propose a mechanism for this reaction. (Hint: This is a condensation because three molecules are joined with loss of water. The mechanism belongs to another class of reactions though.)
Chain branching occurs in cationic polymerization much as it does in free-radical polymerization. Propose a mechanism to show how branching occurs in the cationic polymerization of styrene. Suggest why isobutylene might be a better monomer for cationic polymerization than styrene.
Give the structure of the polyurethane formed by the reaction of toluene diisocyanate
with bisphenol A.
Polypropylene is used in making polymer banknotes and textiles.
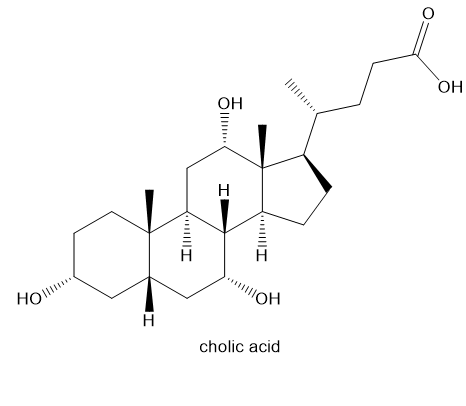
Cholic acid, a major constituent of bile, has the structure shown.
(a) Draw the structure of cholic acid, showing the rings in their chair conformations, and label each methyl group and hydroxy group as axial or equatorial. (Making a model may be helpful.)
(b) Cholic acid is secreted in bile as an amide linked to the amino group of glycine. This cholic acid-amino acid combination acts as an emulsifying agent to disperse lipids in the intestines for easier digestion. Draw the structure of the cholic acid-glycine combination and explain why it is a good emulsifying agent.
What do you think about this solution?
We value your feedback to improve our textbook solutions.