Chapter 26: Q26P (page 1342)
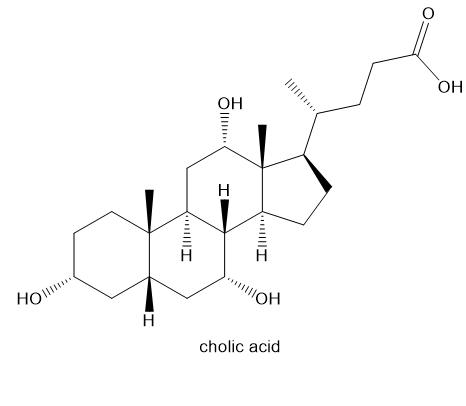
Cholic acid, a major constituent of bile, has the structure shown.
(a) Draw the structure of cholic acid, showing the rings in their chair conformations, and label each methyl group and hydroxy group as axial or equatorial. (Making a model may be helpful.)
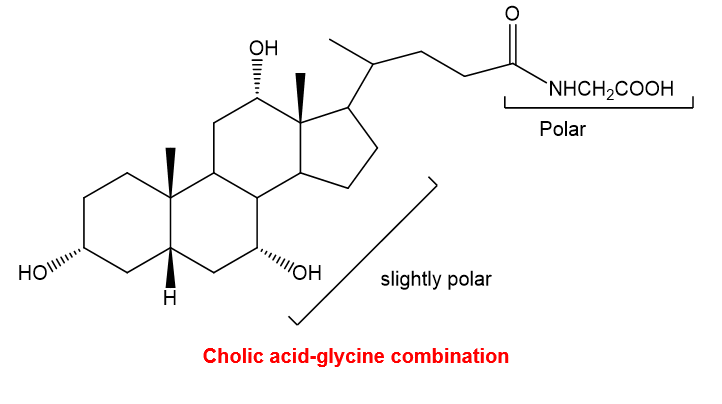
(b) Cholic acid is secreted in bile as an amide linked to the amino group of glycine. This cholic acid-amino acid combination acts as an emulsifying agent to disperse lipids in the intestines for easier digestion. Draw the structure of the cholic acid-glycine combination and explain why it is a good emulsifying agent.
Short Answer