Chapter 26: Q20P (page 1337)
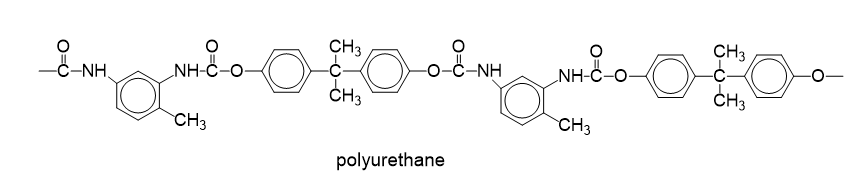
Give the structure of the polyurethane formed by the reaction of toluene diisocyanate
with bisphenol A.
Short Answer
Learning Materials
Features
Discover
Chapter 26: Q20P (page 1337)
Give the structure of the polyurethane formed by the reaction of toluene diisocyanate
with bisphenol A.

All the tools & learning materials you need for study success - in one app.
Get started for free
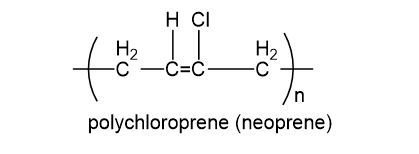
Polychloroprene, commonly known as neoprene, is widely used in wetsuits and in rubber parts that must withstand exposure to gasoline or other solvents.
(a) Is neoprene an addition polymer or a condensation polymer?
(b) What monomer is used to make this synthetic rubber?
Polypropylene is used in making polymer banknotes and textiles.
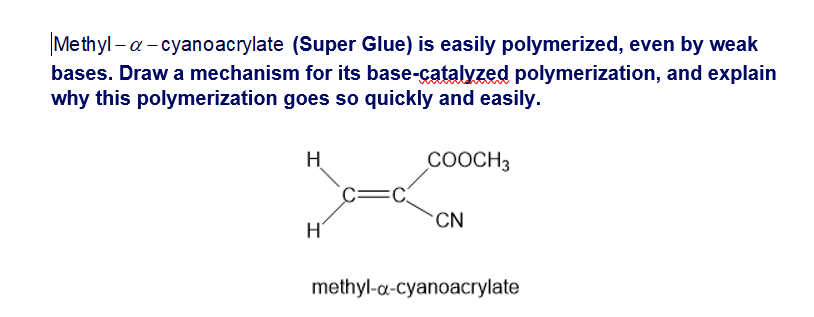
Draw the important resonance forms of the stabilized anion formed in the anionic polymerization of methyl acrylate.
Draw the structure of isotactic poly(acrylonitrile) and syndiotactic polystyrene.
What do you think about this solution?
We value your feedback to improve our textbook solutions.