Chapter 26: Q8P (page 1329)
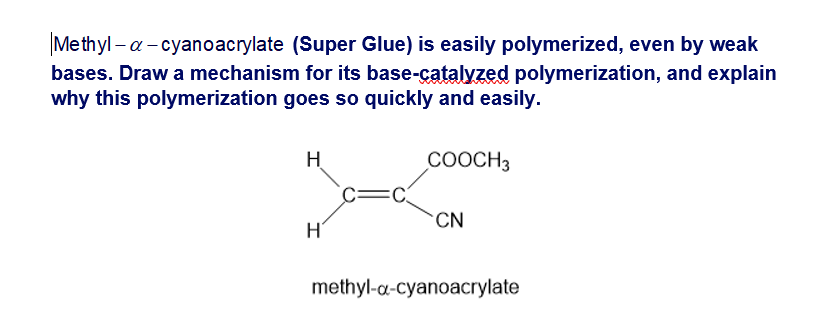
Short Answer
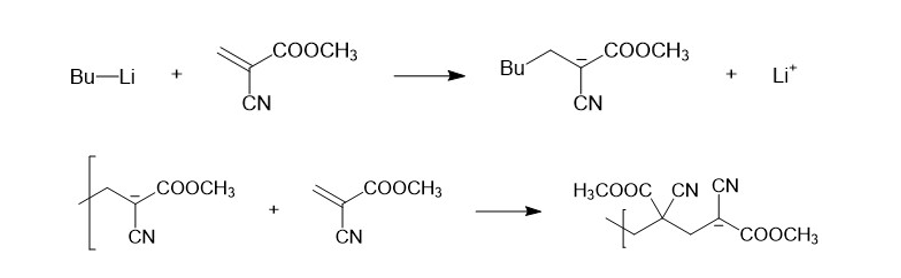
Polymerization goes so quickly and easily because the anion that is formed after deprotonation is stabilized by resonance.
Learning Materials
Features
Discover
Chapter 26: Q8P (page 1329)


Polymerization goes so quickly and easily because the anion that is formed after deprotonation is stabilized by resonance.
All the tools & learning materials you need for study success - in one app.
Get started for free
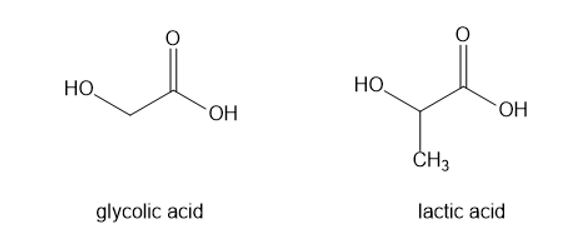
The polyester named LactomerRis an alternating copolymer of lactic acid and glycolic acid. Lactomer is used for absorbable suture material because stitches of Lactomer hydrolyze slowly over a two-week period and do not have to be removed. The hydrolysis products, lactic acid and glycolic acid, are normal metabolites and do not provoke an inflammatory response. Draw the structure of the Lactomer polymer.
Bisphenol A is made on a large scale by a condensation of phenol with acetone. Suggest an appropriate catalyst, and propose a mechanism for this reaction. (Hint: This is a condensation because three molecules are joined with loss of water. The mechanism belongs to another class of reactions though.)
Chain branching occurs in cationic polymerization much as it does in free-radical polymerization. Propose a mechanism to show how branching occurs in the cationic polymerization of styrene. Suggest why isobutylene might be a better monomer for cationic polymerization than styrene.
Compare the molecular structures of cotton and polypropylene, the two major components of thermal underwear. One of these gets wet easily and holds the water in contact with the skin. The other one does not get wet, but wicks the water away from the skin and feels relatively dry to the touch. Explain the difference in how these two fabrics respond to moisture.
Show the intermediate that would result if the growing chain added to the other end of the styrene double bond. Explain why the final polymer has phenyl groups substituted on alternate carbon atoms rather than randomly distributed.
What do you think about this solution?
We value your feedback to improve our textbook solutions.