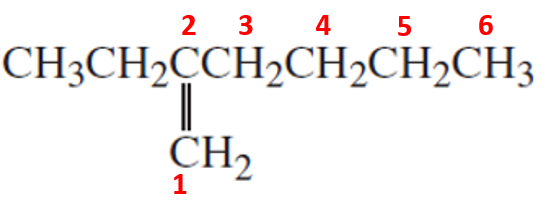Do the numbering of the compounds by identifying the parent chain having maximum number of carbon atoms. Identify the substituents and then name the compound accordingly.
(a)
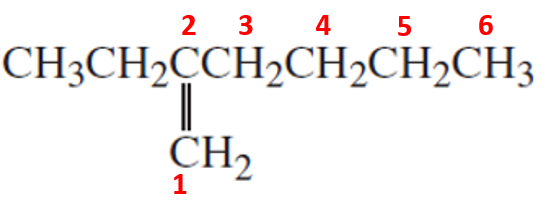
Compound (a)
Here, the parent chain will comprise of 6 carbon atoms and is an alkene. On carbon-2, there is an ethyl substituent. So, the IUPAC name of the compound is 2-ethylhex-1-ene.
(b)
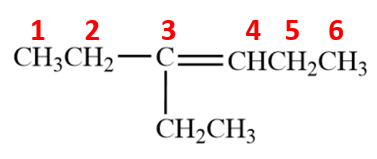
Compound (b)
Here, the parent chain will comprise of 6 carbon atoms and is an alkene. On carbon-3, there is an ethyl substituent. So, the IUPAC name of the compound is 3-ethyl hex-3-ene.
(c)
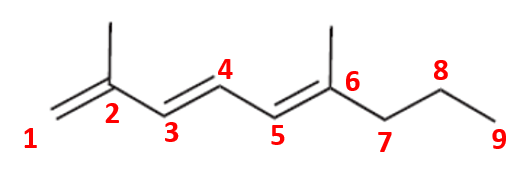
Compound (c)
Here, the parent chain will comprise of 9 carbon atoms and is an alkene. On carbon-2 and carbon-6, there is a methyl substituent. So, the IUPAC name of the compound is 2,6-dimethyl non-1,3,5-triene.
(d)
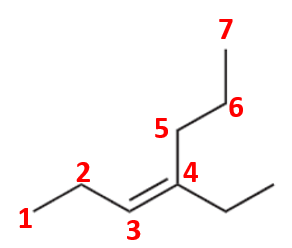
Compound (d)
Here, the parent chain will comprise of 7 carbon atoms and is an alkene. On carbon-4, there is an ethyl substituent. So, the IUPAC name of the compound is 4-ethyl hept-3-ene.
(e)
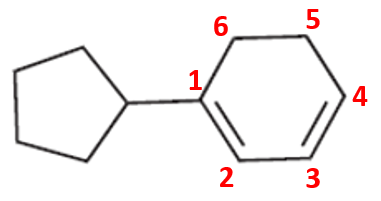
Compound (e)
Here, the parent chain will comprise of 6 carbon atoms and is a cyclic alkene. On carbon-1, there is acyclopentyl substituent. So, the IUPAC name of the compound is 1-cyclopentyl cyclohex-1,3-diene.
(f)
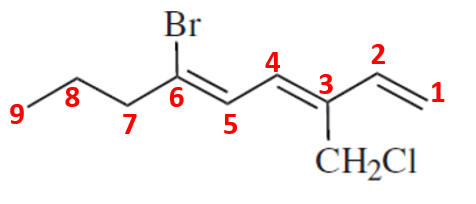
Compound (f)
Here, the parent chain will comprise of 9 carbon atoms and is an alkene with 3 double bonds having the priority. On carbon-3, there is achloromethyl substituent and on carbon-6, there is bromo group as the substituent. So, the IUPAC name of the compound is 6-bromo-3-chloromethyl non-1,3,5-triene.