Chapter 3: Q9P (page 153)
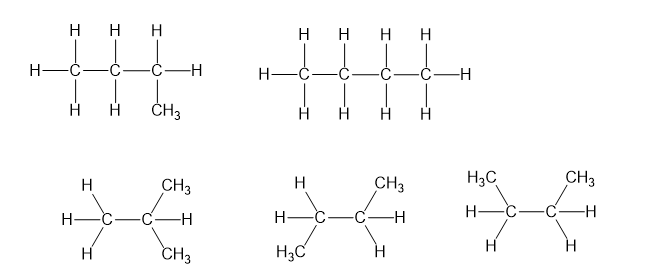
Without looking at the structures, give molecular formulas for the compounds in Problem 3-8 (a) and (b). Use the names of the groups to determine the number of carbon atoms; then use the (2n+2) rule.
Short Answer
(a)C13H28
(b)C16H34