Chapter 3: Q8P (page 153)
Draw the structures of the following compounds.
(a) 4-(1,1-dimethylethyl)nonane
(b) 5-(1,2,2-trimethylpropyl)decane
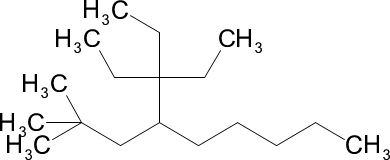
(c) 3,3-diethyl-4-(2,2-dimethylpropyl)nonane
Short Answer
(a)
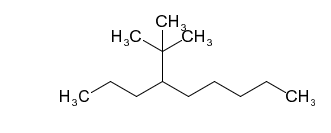
(b)
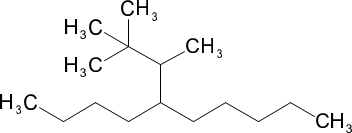
(c)
Learning Materials
Features
Discover
Chapter 3: Q8P (page 153)
Draw the structures of the following compounds.
(a) 4-(1,1-dimethylethyl)nonane
(b) 5-(1,2,2-trimethylpropyl)decane
(c) 3,3-diethyl-4-(2,2-dimethylpropyl)nonane
(a)

(b)

(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
There are eight different five-carbon alkyl groups.
(a) Draw them
(b) Give them systematic names.
(c) In each case, label the degree of substitution (primary, secondary, or tertiary) of the head carbon atom bonded to the main chain.
Name the following alkane and haloalkane. When two or more are present list them in alphabetical order.
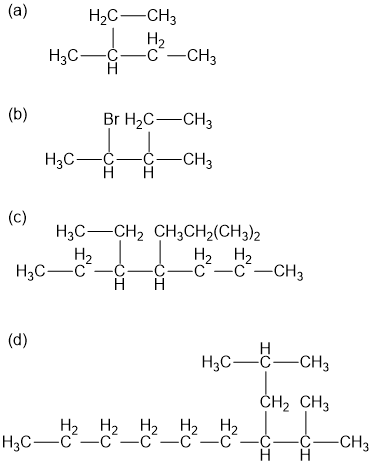
The following names are all incorrect or incomplete, but they represent real structures. Draw each structure and name it correctly.
(a)
(b)
(c)
(d)
(e)
(f)
Draw a perspective representation of the most stable conformation of 3-methylhexane.
Construct a graph, similar to Figure 3-11, of the torsional energy of along the.Placein front, represented by three bonds coming together in a Y shape, andin back, represented by a circle with three bonds pointing out from it. Define the dihedral angle as the angle between the methyl group on the front carbon and ethyl group on the back carbon. Begin your graph at thedihedral angle and begin to turn the front carbon. Show the Newman projection and the approximate energy at eachof rotation. Indicate which conformations are the most stable (lowest energy) and the least stable (highest energy).
What do you think about this solution?
We value your feedback to improve our textbook solutions.