Chapter 3: 46P (page 190)
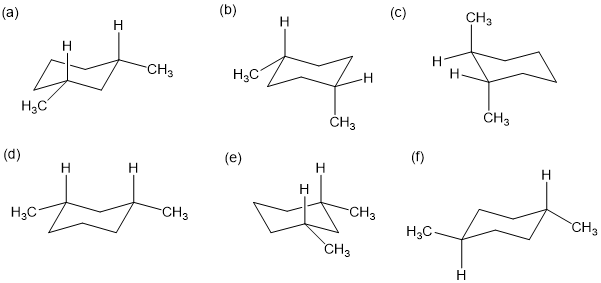
Draw the two chair conformations of each compound, and label the substituents as axial and equatorial. In each case, determine which conformation is more stable.
(a)
(b)
(c)
(d)
(e)
(f)
Learning Materials
Features
Discover
Chapter 3: 46P (page 190)
Draw the two chair conformations of each compound, and label the substituents as axial and equatorial. In each case, determine which conformation is more stable.
(a)
(b)
(c)
(d)
(e)
(f)
All the tools & learning materials you need for study success - in one app.
Get started for free
Name the following compounds. Remember the two up bonds are cis; two down bonds are cis; one up and one down bond are trans.
Question: Conformational studies on ethane-1,2-diol (HOCH2 -CH2OHhave shown the most stable conformation about the central C-Cbond to be the gauche conformation, which is 9.6 KJ/mol (2.3 kcal/mol)more stable than the anti-conformation. Draw Newman projections of these conformers, and explain this curious result.
Draw the structure and give the molecular formula for each of the following compounds.
(a) 1-ethyl-3-methylcycloheptane
(b) isobutylcyclohexane
(c) cyclopropylcyclopentane
(d) 3-ethyl-1,1-dimethylcyclohexane
(e) 3-ethyl-2,4-dimethylhexane
(f) 1,1-diethyl-4-(3,3-dimethylbutyl)cyclohexane
The following names are all incorrect or incomplete, but they represent real structures. Draw each structure and name it correctly.
(a)
(b)
(c)
(d)
(e)
(f)
Question: The heat of combustion of cis-1,2-dimethylcyclopropane is larger than that of trans isomer. Which isomer is most stable? Use drawings to explain this difference in stability.
What do you think about this solution?
We value your feedback to improve our textbook solutions.