Chapter 3: Q19P. (page 170)
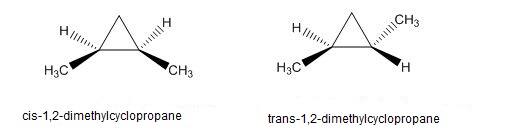
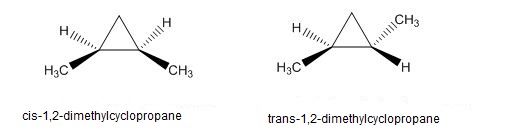
Question: The heat of combustion of cis-1,2-dimethylcyclopropane is larger than that of trans isomer. Which isomer is most stable? Use drawings to explain this difference in stability.
Short Answer
Answer
Between cis-1,2-dimethylcyclopropane and trans-1,2-dimethylcyclopropane, trans-1,2-dimethylcyclopropane is more stable.