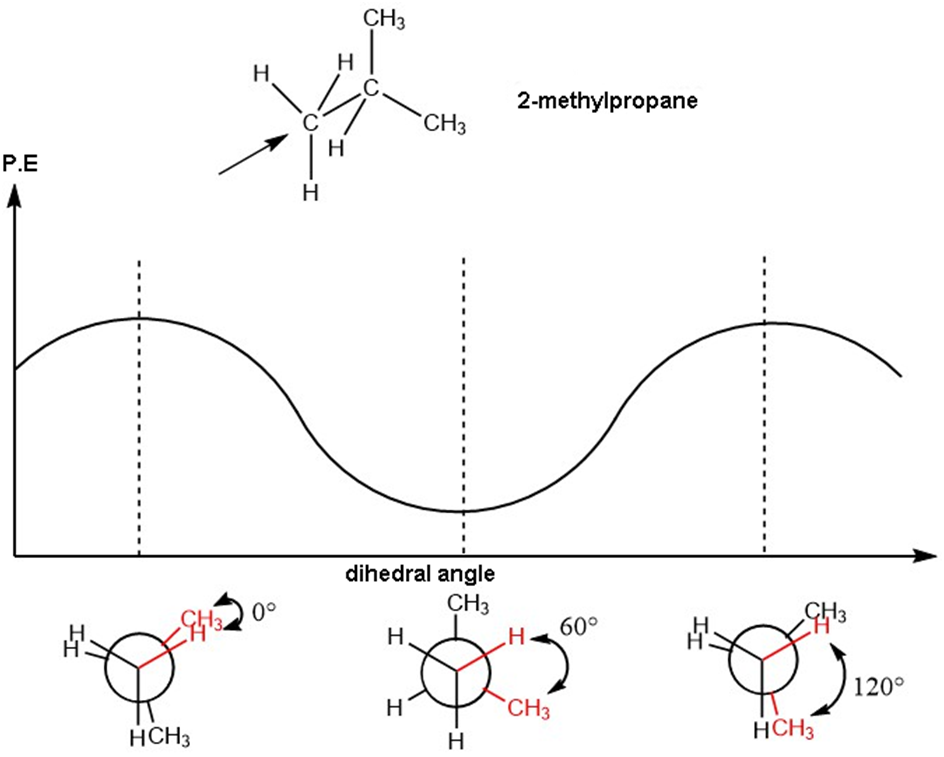
Chapter 3: Q12P (page 161)
Question: Draw a graph, similar to Figure 3-9, of the torsional strain of as it rotates about the bond between and . Show the dihedral angle and draw a Newman projection for each staggered and eclipsed conformation.
Short Answer