Chapter 3: Q13P (page 164)
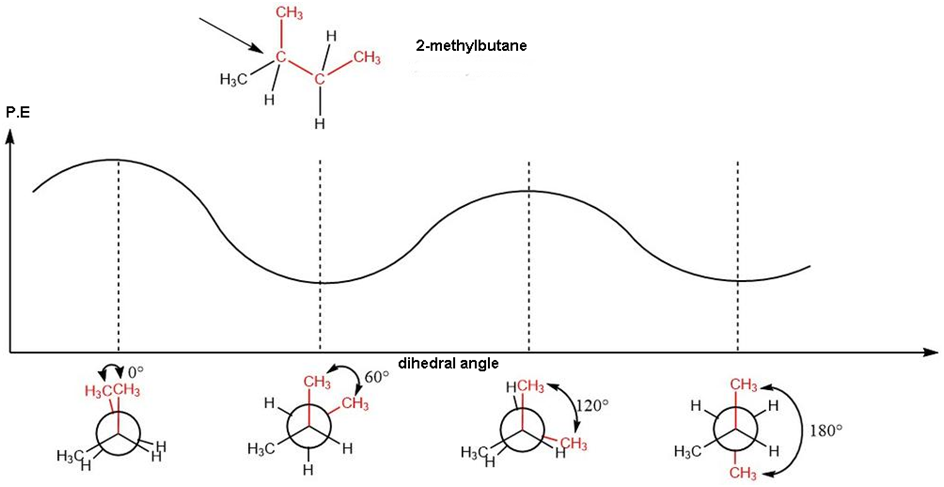
Question: Draw a graph, similar to Figure 3-11, of the torsional energy of as it rotates about the bond.
Short Answer
Learning Materials
Features
Discover
Chapter 3: Q13P (page 164)
Question: Draw a graph, similar to Figure 3-11, of the torsional energy of as it rotates about the bond.

All the tools & learning materials you need for study success - in one app.
Get started for free
(a) Draw and name the five cycloalkane structures of formula. Can any of these structures give rise to geometric (cis-trans) isomerism? If so, show the cis and trans stereoisomers.
(b) Draw and name the eight cycloalkane structures of formulathat do not show geometric isomerism.
(c) Draw and name the four cycloalkanes of formulathat do have cis-trans isomerism.
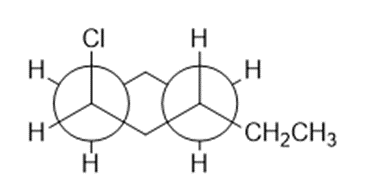
Question: This is a Newman projection of a substituted cyclohexane.
(a) Draw the equivalent chair form
(b)Draw the equivalent structure using wedge and dash notation on a cyclohexane hexagon.
(c) Give the IUPAC name.
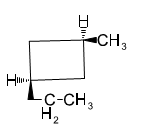
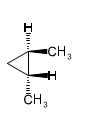
Give IUPAC names for the following cycloalkanes.
(a)
(b)

(c)
Name the following alkane and haloalkane. When two or more are present list them in alphabetical order.
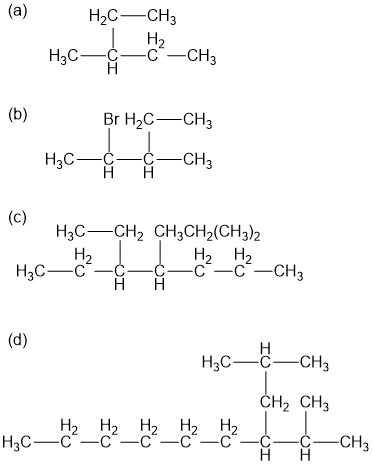
In each pair of compounds, which compound has the higher boiling point? Explain your reasoning.
(a) Nonane or 3-methylheptane
(b) Octane or 2,3,4-trimethylpentane
(c) 2,3,5-trimethylhexane or nonane
What do you think about this solution?
We value your feedback to improve our textbook solutions.